What is a pi molecular orbital?
What is a pi molecular orbital?
Pi orbital (π orbital): The bonding molecular orbital component of a pi bond. The π orbital of ethylene’s carbon-carbon pi bond has two orbital lobes, one above the plane of the atoms, and another below the plane. This is a bonding molecular orbital. The plane containing the atoms is also the pi orbital’s one node.
How many pi molecular orbitals are there?
four pi molecular orbitals
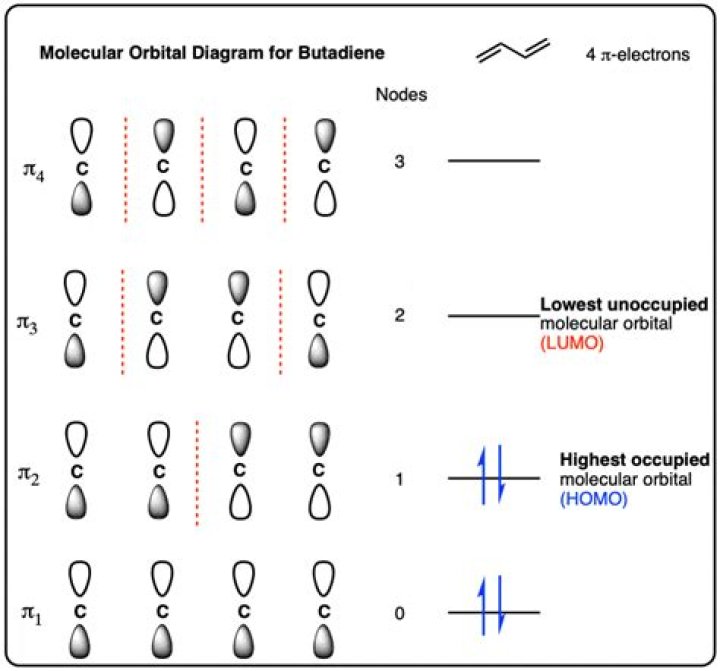
There are four adjacent carbon atoms involved in the pi system and the combination of a p orbital from each of these atoms will result in four pi molecular orbitals: ψ1, ψ2, ψ3*, and ψ4* (also referred to as π1, π2, π3*, and π4*).
Which molecular orbital is highest in energy pi or Sigma?
pi molecular orbital
The sigma bond between the two carbon atoms does not have a node in the plane of the molecule. The pi bond between the two carbon atoms has one node in the plane of the molecule. Thus the pi molecular orbital is higher in energy and is the highest occupied molecular orbital (the HOMO).
Do molecular orbitals involve pi bonding?
Molecular orbitals involve pi bonding. A bonding orbital is a molecular orbital whose energy is hight than that of the atomic orbitals from which it is found. With hybridization, several atomic orbitals overlap to form the same total number of equivalent hybrid orbitals.
What is pi and pi * orbitals?
The corresponding antibonding, or π* (“pi-star”) molecular orbital, is defined by the presence of an additional nodal plane between these two bonded atoms.
How many pi electrons does guanine?
ten pi electrons
Indole (a functional group on the side chain of the amino acid tryptophan) and purine (found in guanine and adenine nucleotide bases) both have a total of ten pi electrons delocalized around two rings.
Why is pi bond weaker than sigma?
In a π bond, the p orbitals overlap side-on. The overlap is less efficient, because the electron density is off to the sides of the σ bond. The electrons are not as effective in attracting the two nuclei. Thus, a π bond is weaker than a σ bond.
Are pi orbitals lower in energy than sigma orbitals?
The reason for this is that the atomic orbital overlap and thus the strength of the interaction are greater for a σ bond than a π bond, which means that the σ molecular orbital is more stable (lower in energy) than the π molecular orbitals.


