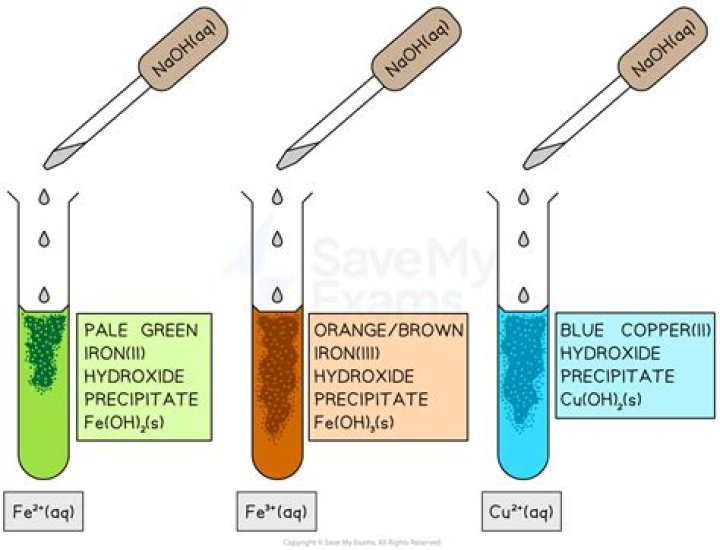
Hydroxide ion (OH-) binds to the copper (II) ion even more strongly than does water. As a result, hydroxide ion can displace water from the copper (II) ion, yielding copper hydroxide, Cu(OH)2, a blue precipitate. Heating copper hydroxide produces copper oxide, CuO, a black solid..
Similarly one may ask, is copper hydroxide soluble or insoluble in water?
Based on the solubility rules copper hydroxide is not soluble in water while copper chloride is. Ksp is very small meaning that copper hydroxide dissociates very little in water, while the vast majority remains a solid.
is copper hydroxide a solid? Copper hydroxide is a pale blue solid. Some forms of copper(II) hydroxide are sold as "stabilized" copper hydroxide, quite likely a mixture of copper(II) carbonate and hydroxide. These are often greener in colour. Copper hydroxide acts as a weak base in aqueous solution.
Likewise, people ask, is copper chloride a precipitate?
A light blue precipitate forms when sodium carbonate reacts with copper(II) chloride. No precipitate forms when sodium sulphate reacts with copper(II) chloride.
What does copper form a precipitate with?
Copper(II) ion reacts with stoichiometric quantities of aqueous ammonia to precipitate light blue Cu(OH)2. Some basic salts may also form. The precipitate does not dissolve in excess sodium hydroxide unless the NaOH solution is very concentrated.
Related Question Answers
Why is copper oxide insoluble in water?
Copper oxide is a base because it will react with acids and neutralise them, but it is notan alkali because it does not dissolve in water. Sodium hydroxide is a base because it will react with acids and neutralise them. It's also an alkali because it dissolves in water.Is copper hydroxide toxic?
Local toxicity But, according to the requirements specified by Directive 67/548/EC and subsequent regulations, copper hydroxide is not classified as a skin irritant.Is copper hydroxide a weak base?
It is a pale greenish blue or bluish green solid. Some forms of copper(II) hydroxide are sold as "stabilized" copper(II) hydroxide, although they likely consist of a mixture of copper(II) carbonate and hydroxide. Cupric hydroxide is a weak base.Is copper soluble in water?
The pure compound is stable in dry air. Moist air converts it to a greenish oxygenated compound, and upon exposure to light it is transformed into copper(II) chloride. It is insoluble in water but dissolves in concentrated hydrochloric acid or in ammonia because of the formation of complex ions.Is copper chloride soluble in water?
Water
Which hydroxide is soluble in water?
Alkali earth metals Only strontium hydroxide ( Sr(OH)2) and barium hydroxides ( Ba(OH)2 ) are completely soluble from alkaline earth metals. Berrylium hydroxide ( Be(OH)2) and magnesium hydroxide ( Mg(OH)2) are completely insoluble in water.What is copper hydroxide used for?
Copper(II) hydroxide is used to kill mold in paints. It can be used to color ceramics. It can be used as a catalyst.Why does copper chloride dissolve in water?
Yes, copper (II) chloride does dissolve in water. In order to figure out if any of its parts hydrolyzes or not, we have to break down the salt first. The (II) in copper (II) indicates that the copper atom has a charge of , while chloride, which is basically chlorine as an anion, always have a charge of a single .What Colour is copper chloride solution?
blue
What color is copper chloride hydrate when heated?
Copper(II) Chloride (CuCl2) In its anhydrous state, CuCl2 is brown in color. Anhydrous copper chloride is hygroscopic and will absorb moisture from the air to form the dihydrate. By heating the hydrated copper chloride, one can drive off the water from the CuCl2 • 2 H2O crystals to form anhydrous CuCl2.What happens if you drink copper chloride?
* Repeated exposure can cause thickening of the skin. * Breathing Copper Chloride can irritate the nose, throat and lungs causing coughing and wheezing. * Copper Chloride can irritate the stomach causing salivation, nausea, vomiting, stomach pain and diarrhea. * Copper Chloride may damage the liver and kidneys.What is copper chloride made of?
Copper(II) chloride is the chemical compound with the chemical formula CuCl2. This is a light brown solid, which slowly absorbs moisture to form a blue-green dihydrate.How is copper chloride produced?
Copper chloride is formed when copper reacts with chlorine radicals. It is probably a photochemical reaction, where the sunlight provides the energy to break the bonds of chlorine gas to form chlorine radicals. How do you precipitate copper?
Hydroxide ions (from, say, sodium hydroxide solution) remove hydrogen ions from the water ligands attached to the copper ion. Once a hydrogen ion has been removed from two of the water molecules, you are left with a complex with no charge - a neutral complex. This is insoluble in water and a precipitate is formed.Is copper chloride a solution?
Cupric chloride. Description: Cupric chloride, for injection, is a sterile, nonpyrogenic solution intended for use as an additive to solutions for Total Parenteral Nutrition (TPN). Copper chloride appears as a yellowish-brown powder (the anhydrous form) or a green crystalline solid (the dihydrate).How do you precipitate chloride?
A precipitate of silver chloride is formed by adding a solution of silver nitrate to the aqueous solution of chloride ions. The precipitate is collected by careful filtration and weighed. The precipitate can be collected more easily if the reaction solution is heated before filtering.What is the formula for copper 2 hydroxide?
Cu(OH)2
Why is copper hydroxide a solid?
As a result, hydroxide ion can displace water from the copper (II) ion, yielding copper hydroxide, Cu(OH)2, a blue precipitate. Heating copper hydroxide produces copper oxide, CuO, a black solid. At the same time, some of the zinc metal, which is present in excess, reduces hydronium ions to H2.Why does zinc turn black in copper sulfate?
When a strip of zinc metal is placed into a blue solution of copper(II) sulfate ( Figure below ), a reaction immediately begins as the zinc strip begins to darken. If left in the solution for a longer period of time, the zinc will gradually decay due to oxidation to zinc ions.