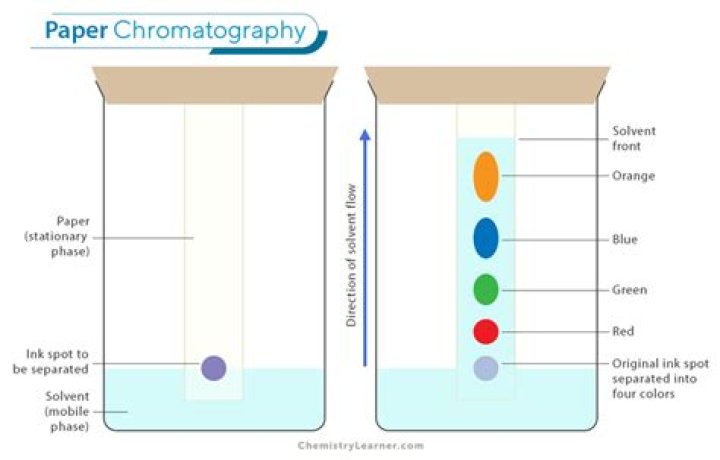
Paper chromatography uses paper as the stationary phase. The exact type of paper used is important. Filter paper is one of the best types, although paper towels and even newspaper can also be used. Writing paper is coated so that ink does not run and because of this is less satisfactory..
In this regard, why do we use filter paper for chromatography?
You place your filter paper in the sealed development chamber after the solvent but before the stationary phase. It absorbs the liquid in the solvent and provides more surface area for evaporation. More surface area means more and faster evaporation.
Furthermore, what can I use instead of chromatography paper? Porous household papers like paper towels and coffee filters make an inexpensive substitute for chromatography paper.
Just so, what solvent is used in paper chromatography and how does it work?
Paper chromatography using a water and other polar solvents All substances should be equally soluble (or equally insoluble) in both. And yet the first chromatograms that you made were probably of inks using water as your solvent.
How do you choose a solvent for paper chromatography?
Criteria for selection of a working solvent system
- Solvents should not be toxic or carcinogenic.
- The solvent mixture composition should not change with time.
- Solvent constituents should not chemically react with any of the sample constituents.
Related Question Answers
What is the best solvent for paper chromatography?
Readily Available Solvents for Paper Chromatography
| Solvent | Polarity (arbitrary scale of 1-5) | Suitability |
| Water | 1 – Most polar | Good |
| Rubbing alcohol (ethyl type) or denatured alcohol | 2 – High polarity | Good |
| Rubbing alcohol (isopropyl type) | 3 – Medium polarity | Good |
| Vinegar | 3 – Medium polarity | Good |
What is the basic importance of chromatography?
Chromatography is used for quality analyses and checker in the food industry, by identifying and separating, analyzing additives, vitamins, preservatives, proteins, and amino acids. Chromatography like HPLC is used in DNA fingerprinting and bioinformatics.What is the basic principle of paper chromatography?
Principle of paper chromatography: The principle involved is partition chromatography wherein the substances are distributed or partitioned between liquid phases. One phase is the water, which is held in the pores of the filter paper used; and other is the mobile phase which moves over the paper.What is the purpose of filter paper?
Filter paper is a semi-permeable paper barrier placed perpendicular to a liquid or air flow. It is used to separate fine substances from liquids or air. It is used in science labs to remove solids from liquids. This can be used to remove sand from water.Why do the colors separate in paper chromatography?
As the water creeps up the paper, the colors will separate out into their components. Capillary action makes the solvent travel up the paper, where it meets and dissolves the ink. The dissolved ink (the mobile phase) slowly travels up the paper (the stationary phase) and separates out into different components.What is an RF value?
RF value (in chromatography) The distance travelled by a given component divided by the distance travelled by the solvent front. For a given system at a known temperature, it is a characteristic of the component and can be used to identify components.What type of paper is used for chromatography?
Paper chromatography uses paper as the stationary phase. The exact type of paper used is important. Filter paper is one of the best types, although paper towels and even newspaper can also be used. Writing paper is coated so that ink does not run and because of this is less satisfactory.Why are two solvents used in chromatography?
Chromatography is a technique used to separate the components of a mixture. Different solvents will dissolve different substances. A polar solvent (water) will dissolve polar substances (water soluble ink in the video below). A non-polar solvent will dissolve non-polar substances.Why is water not a suitable solvent in paper chromatography?
Technically it can, but it almost always isn't because the idea is to seperate out compounds based on their solubility in different types of solvents. Cellulose (paper) is polar and water is obviously quite polar, so there's no difference there by which to seperate out the compounds.What is the RF value in paper chromatography?
The Rf value is defined as the ratio of the distance moved by the solute (i.e. the dye or pigment under test) and the distance moved by the the solvent (known as the Solvent front) along the paper, where both distances are measured from the common Origin or Application Baseline, that is the point where the sample isWhat factors affect paper chromatography?
Retention factor values in thin layer chromatography are affected by the absorbent, the solvent, the chromatography plate itself, application technique and the temperature of the solvent and plate.Which solvent is more soluble in chromatography?
Based on the Rf values, xanthophylls are more soluble in the chromatography solvent.What are the advantages of paper chromatography?
The main advantages that paper chromatography offers are simplicity, low cost, and unattended, hassle-free operation. It can be run in various modes, and quantitation may be achieved without the use of expensive instrumentation.Why is acetone a good solvent for chromatography?
Its slight polarity allows it to dissolve polar substances, and the fact that it is less polar than water allows greater resolution between pigments on paper. These reasons allow acetone to be a great solvent for pigment chromatography. Other solvents that are good are small alcohols for the same reason!What mixtures can be separated by chromatography?
Separating dissolved solids – chromatography. Paper chromatography is a method for separating dissolved substances from one another. It is often used when the dissolved substances are coloured, such as inks, food colourings and plant dyes.What solvent is used in column chromatography?
The two most common stationary phases for column chromatography are silica gel (SiO2) and alumina (Al2O3), with the most commonly used mobile phases being organic solvents. The solvent(s) chosen for the mobile phase are dependent on the polarity of the molecules being purified.What is stationary phase in paper chromatography?
In paper chromatography, substances are distributed between a stationary phase and a mobile phase. The stationary phase is the water trapped between the cellulose fibers of the paper. The mobile phase is a developing solution that travels up the stationary phase, carrying the samples with it.What happened to the ink in each solvent?
When ink is exposed to certain solvents the colors dissolve and can be seperated out. When we expose a piece of paper with ink on it to a solvent, the ink spreads across the paper when the ink dissolves. Some inks are water-soluble, so you can use water as the solvent.Why is alcohol used in chromatography?
So the only reason why you use alcohol rather than water is because your ink is soluble in ethanol but not in water. In this experiment, rubbing alcohol (isopropyl alcohol) is used as the solvent (mobile phase) for paper chromatography separation.