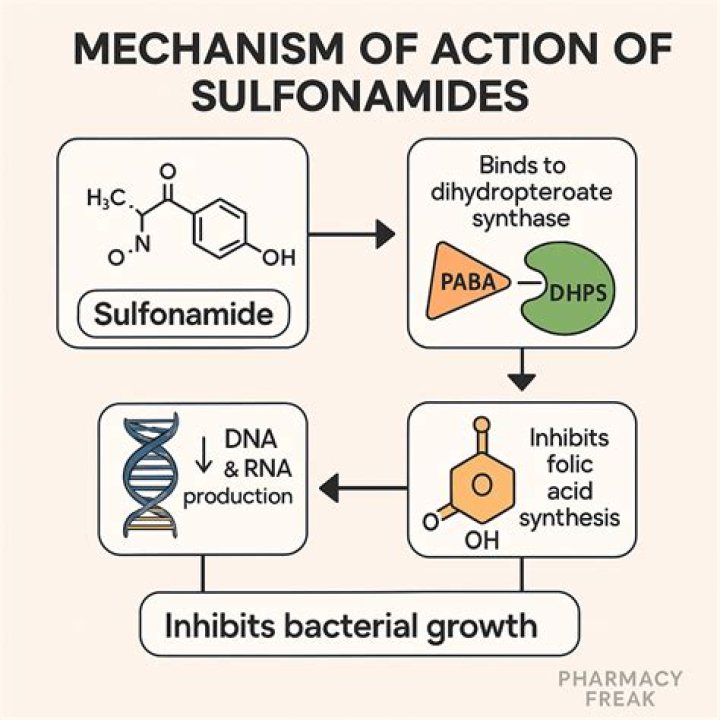
Why is the SRK equation of state called a cubic equation of state?

Why is the SRK equation of state called a cubic equation of state?
Cubic equations of state. These equations are called “cubic” because they are of the third degree in molar volume, i.e. (3.1-15) (3.1-16)
What is the formula for critical temperature?
Tc=27Rb8a.
How do you find B in virial equation?
The value of B’ can be calculated by dividing the slope by the intercept: B’ = slope/intercept . The virial coefficient B may be calculated by multiplying B’ by RT, and the number of moles in the sample (n) may be calculated by dividing the intercept by RT.
What do you mean by equation of state?
In physics and thermodynamics, an equation of state is a thermodynamic equation relating state variables which describe the state of matter under a given set of physical conditions, such as pressure, volume, temperature (PVT), or internal energy.
How use Shahrukh Khan equation?
To use the SRK equation:
- look up Tc, Pc, and the acentric factor.
- plug in and find a, b, and alpha.
- plug these into the SRK equation; the result will be a cubic equation in P, T, and V.
- solve for the unknown you seek.
What is the Peng-Robinson equation of state?
The Peng-Robinson equation of state was used to calculate the volume of 100% methane gas as a function of pressure and temperature (Peng and Robinson, 1976). This equation expresses fluid properties in terms of the critical properties and acentric factor of each species involved.
What is a reduced equation of state?
an equation relating the reduced parameters of a thermodynamic equilibrium system, that is, the parameters of pressure, volume, and temperature, defined as the ratio of their values to the critical state.
What is Wonderwall equation of state?
The van der Waals equation is an equation of state that corrects for two properties of real gases: the excluded volume of gas particles and attractive forces between gas molecules. The van der Waals equation is frequently presented as: (P+an2V2)(V−nb)=nRT ( P + a n 2 V 2 ) ( V − n b ) = n R T .
What is virial equation state?
The virial Equation of state is a model that attempts to describe the properties of a real gas. This temperature is known as the Boyle temperature, TB, and it is the temperature at which the repulsive forces between the gas molecules exactly balance the attractive forces between the gas molecules.
Which equation is equation of state?
In the limit of low pressures and high temperatures, where the molecules of the gas move almost independently of one another, all gases obey an equation of state known as the ideal gas law: PV = nRT, where n is the number of moles of the gas and R is the universal gas constant, 8.3145 joules per K.
What is the most accurate equation of state?
In physics and thermodynamics, the Redlich–Kwong equation of state is an empirical, algebraic equation that relates temperature, pressure, and volume of gases. It is generally more accurate than the van der Waals equation and the ideal gas equation at temperatures above the critical temperature.