Which is more polar phenacetin or aspirin?
Which is more polar phenacetin or aspirin?
Aspirin has the third highest R F value with two dipole-dipole forces and one hydrogen bond, almost identical to the R F value of Phenacetin. Acetaminophen has two hydrogen bonds and one dipole-dipole making it more polar than Aspirin, Phenacetin, and Ibuprofen.
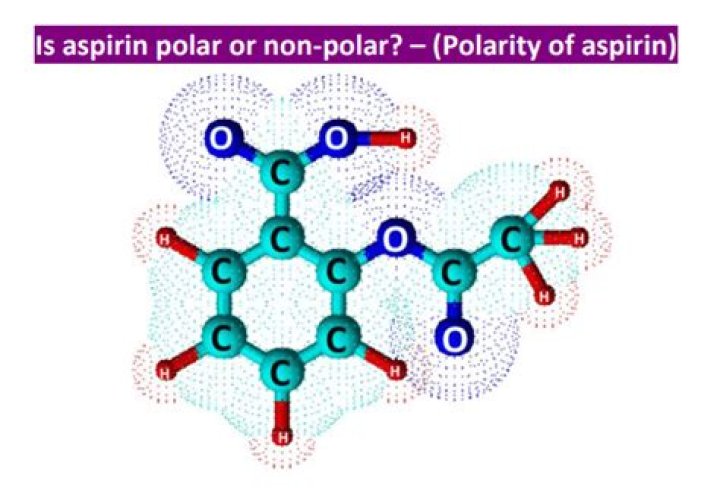
Is aspirin nonpolar or polar?
Aspirin is a polar molecule with dipole-dipole attraction bonds and an -OH (hydroxyl) segment as part of a carboxylic acid group. This makes it easily dissoluble in other polar liquids, such as water (H2O) and blood plasma.
Is aspirin acetaminophen or caffeine more polar?
Acetaminophen has a phenol and amide function group, but caffeine has multiple amide functional groups; therefore, acetaminophen is more polar than aspirin and less polar than caffeine.
Which of the following compounds is most polar acetaminophen ibuprofen aspirin caffeine?
Ibuprofen is least polar and Caffeine is most polar. caffeine contains two amide groups, two amine groups and an alkene group.
Is phenacetin soluble in water?
Phenacetin is significantly less soluble in hot water (1.22 grams per 100 mL of boiling water) than acetanilide is (5.0 grams per 100 mL of boiling water).
How do you determine polarity?
The terms “polar” and “nonpolar” usually refer to covalent bonds. To determine the polarity of a covalent bond using numerical means, find the difference between the electronegativity of the atoms; if the result is between 0.4 and 1.7, then, generally, the bond is polar covalent.
Is Tylenol polar or nonpolar?
Based on the rate of migration, one can conclude that the acetaminophen, aspirin and caffeine are polar and prefer to be on the stationary phase while the naproxen sodium, unknown and ibuprofen are less polar with weak bonds.
Is acetaminophen soluble in water?
Water
AcetoneAlcohol
Acetaminophen/Soluble in
Is acetaminophen soluble in dichloromethane?
Because they are insoluble in dichloromethane, the binder and acetaminophen will be separated from the other two components by treatment of the solid with hot dichloromethane. The acetaminophen is soluble in hot ethanol. The other two components can then be separated using a standard acid/base extraction.
What is phenacetin soluble in?
Phenacetin occurs at room temperature as white, odorless monoclinic prisms. It is soluble in water (more so in hot than cold water), alcohol, glycerol, and acetone and is slightly soluble in benzene.
How do you find polarity without electronegativity?
To review the steps:
- Draw the Lewis structure.
- Figure out the geometry (using VSEPR theory)
- Visualize or draw the geometry.
- Find the net dipole moment (you don’t have to actually do calculations if you can visualize it)
- If the net dipole moment is zero, it is non-polar. Otherwise, it is polar.