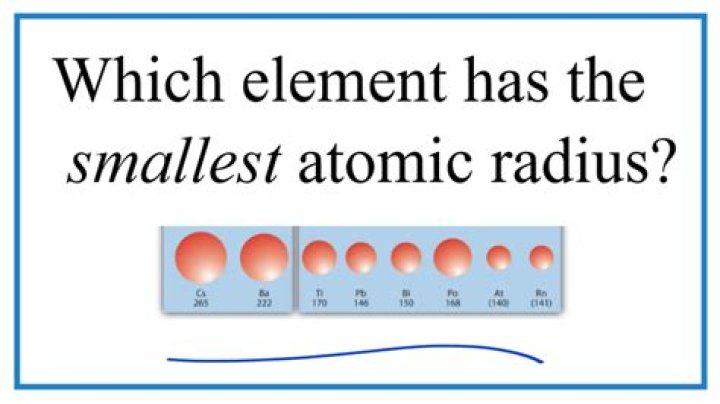
Helium
.
Similarly one may ask, which element has the smallest volume?
The element with smallest radii is Helium.
Also, is helium the smallest atom? Helium has two protons and two electrons in the same shell as hydrogen . Hydrogen has that 1 electron in k shell and helium has 2 electrons in k shell . Therefore helium has the smallest atomic size .
In this regard, which has smallest size?
Helium has the smallest atomic radius because the stronger force between the two protons and two electrons pulls the electron shell in closer than the lone electron and lone proton in a Hydrogen atom.
Which element has the highest ionization energy?
From this trend, Cesium is said to have the lowest ionization energy and Fluorine is said to have the highest ionization energy (with the exception of Helium and Neon).
Related Question Answers
Why are cations smaller?
Cations and Anions These cations are smaller than their respective atoms; this is because when an electron is lost, electron-electron repulsion (and therefore, shielding) decreases and the protons are better able to pull the remaining electrons towards the nucleus (in other words, Zeffincreases).Does Al or B have a larger atomic radius?
Originally Answered: Among elements Al Si B and C which has the largest atomic radius? Aluminium Al will have the largest atomic radius, considering the trends that atomic radius decreases across a period and increases down a group.Which element has the smallest atomic radius?
As can be seen in the figures below, the atomic radius increases from top to bottom in a group, and decreases from left to right across a period. Thus, helium is the smallest element, and francium is the largest.Where is the atomic radius on an element?
The atomic radius of a chemical element is the distance from the centre of the nucleus to the outermost shell of the electron.What has a larger radius than zinc?
Atomic Radius of the elements
| Helium | 31 pm | Praseodymium |
| Antimony | 133 pm | Seaborgium |
| Polonium | 135 pm | Bohrium |
| Gallium | 136 pm | Hassium |
| Zinc | 142 pm | Meitnerium |
Why does oxygen have a larger atomic radius than nitrogen?
The eight protons in oxygen atom's nucleus will exert more force than nitrogen's seven electrons. Also, oxygen has same number of shells as nitrogen. Thus electrons in oxygen atom will be closer to nucleus than in the case of nitrogen. When electrons are farther away from nucleus atomic radius is also larger.Which metal is smallest in size?
Beryllium
What is the size of an element?
As you move down an element group (column), the size of atoms increases. This is because each atom further down the column has more protons and neutrons and also gains an additional electron energy shell. As you move across an element period (row), the overall size of atoms decreases slightly.What is the largest element?
This periodic table shows the relative sizes of the atoms of each element. Each atom's size is relative to the largest element, cesium.Which element has largest atom?
Francium
Which is the most electronegative atom?
Fluorine
Which element has biggest atomic size?
Cesium
How do you determine electronegativity?
To calculate electronegativity, start by going online to find an electronegativity table. You can then assess the quality of a bond between 2 atoms by looking up their electronegativities on the table and subtracting the smaller one from the larger one. If the difference is less than 0.5, the bond is nonpolar covalent.Whats is an atom?
An atom a fundamental piece of matter. An atom itself is made up of three tiny kinds of particles called subatomic particles: protons, neutrons, and electrons. The protons and the neutrons make up the center of the atom called the nucleus and the electrons fly around above the nucleus in a small cloud.What is meant by atomic radius?
The atomic radius of a chemical element is a measure of the size of its atoms, usually the mean or typical distance from the center of the nucleus to the boundary of the surrounding shells of electrons. The value of the radius may depend on the atom's state and context.Which element has the smallest electronegativity?
Electronegativity increases moving from the bottom left-hand corner of the periodic table toward the upper right-hand corner. The element with the lowest electronegativity value is francium, which has an electronegativity of 0.7. This value uses the Pauling scale to measure electronegativity.Which is smaller H or he?
Helium has an atomic radius of 31 pm, hydrogen has an atomic radius of about 53 pm. So an atom of helium is significantly smaller than an atom of hydrogen measuring by the radius of the electron cloud. This is mostly because the charge of the helium nucleus is twice as big as that of the hydrogen nucleus.How big can an atom get?
Atoms are composed of a nucleus (where the positively charged protons and uncharged neutrons reside) surrounded by a cloud of orbiting negatively charged electrons. An atom is about 10-8 centimeters in size (meaning that 100 million of them would fit side-by-side within one centimeter).Why is the atomic radius of k larger than na?
As an example, potassium (K) has a larger average atomic radius (220 pm)than sodium (Na) does (180 pm). The potassium atom has an extra electron shell compared to the sodium atom, which means its valence electrons are further from the nucleus, giving potassium a larger atomic radius.