Where does the heat go as the hot chocolate cools down?
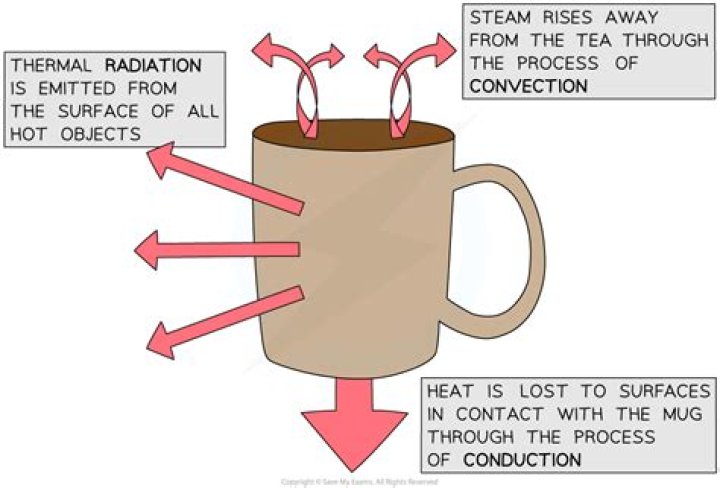
Where does the heat go as the hot chocolate cools down?
Heat Transfer Heat flows from a hotter location (more energy) to a cooler one (less energy). For example, if you add cold milk to your hot chocolate, heat flows from the hot chocolate to the cold milk. The hot chocolate cools because heat flows away; the cold milk warms because heat flows in.
Why does a mug of hot chocolate eventually cool down?
If the mug is initially cold, some heat will be transferred to get it to the same temperature as the liquid; after that, the mug will cool due to contact with air (and a tiny bit due to thermal radiation), and this will set up an equal flow of heat from the liquid to the mug in the steady state.
When you pour hot cocoa into a cool mug what happens?
When you pour cold milk into hot cocoa, the milk and cocoa particles start to collide When a high-energy cocoa particle hits a low-energy milk particle, energy transfers. The cocoa particles slow down, and the cup of cocoa cools down. Look at the illustration on page 33.
What causes the hot chocolate effect?
Mixing hot chocolate mix and hot water creates bubbles on the surface. Stirring them together causes the bubbles to spread out through the liquid. The speed of sound travels slower in air bubbles than it does through a liquid. With the bubbles mixed into the liquid, the pitch becomes lower.
How do you know when the transfer of heat has ended?
How do you know when the transfer of thermal energy has ended? Thermal energy never stops transferring. When one substance begins to boil. When you can measure the amount of heat.
What is wrong with this statement a mug of hot chocolate contains more heat than a glass of cold water?
Explain what is wrong with the statement: “A mug of hot chocolate contains more heat than a glass of cold water.” energy between warm and cool particles. The correct way to state this statement is “A mug of hot chocolate contains more energy than a glass of cold water.”
How is heat lost from hot water?
Evaporation is the process of losing heat through the conversion of water to gas (evaporation of sweat). The primary heat loss process for aqua enthusiasts is convection, however, in an outdoor pool on hot day evaporation will also play a primary role in heat loss.
What is wrong with the statement a mug of hot chocolate contains more heat than a glass of cold water?
What happen when you mix powdered chocolate in a hot water?
When you mix cocoa into water or milk, it creates a fine foam made of tiny bubbles on the surface. Vigorous stirring spreads the bubbles throughout the liquid. The bubbles are filled with air, of course, and sound travels much slower through air than through the liquid.
Is mixing hot chocolate mix into a cup of water a chemical reaction?
Making hot chocolate may be categorized as a chemical reaction. Almost everything that occurs around us is considered to have a chemical reaction….
How does a hot chocolate mug cool itself down?
If a hot cup of hot chocolate is just standing there, can it cool itself down by transferring the kinetic/thermal energy that the liquid has into the mug/cup? There’s a strong dependence of this phenomenon on the substance. Some hot chocolates take longer to cool than others.
What happens when you mix hot and cold chocolate?
As soon as you mix the chocolate powder with the boiling water, you take a sip and burn your tongue because it’s too hot. You decide to let it cool down for 10 minutes. But, when you sip the hot chocolate 10 minutes later, it’s too cold—and no one likes a cup of cold chocolate!
How is the cooling rate of hot chocolate determined?
They collect and graph data to create a mathematical model that represents the cooling rate, and use an exponential decay regression to determine how long a person should wait to drink the cup of hot chocolate at an optimal temperature. In Part 2, students investigate the specific heat capacity of the hot chocolate.
What can I do with a cup of hot chocolate?
This activity also uses some non-expendable (reusable) items such as Arduino® microcontrollers and temperature sensors, although they are optional; simple thermometers may be used instead. A cup of hot chocolate with an Arduino.
If a hot cup of hot chocolate is just standing there, can it cool itself down by transferring the kinetic/thermal energy that the liquid has into the mug/cup? There’s a strong dependence of this phenomenon on the substance. Some hot chocolates take longer to cool than others.
What happens to hot chocolate when it goes bad?
Hot chocolate that has gone bad does not have its original sweet aroma. The hot chocolate turns lumpy due to the presence of water. Don’t use the hot chocolate powder if it seems wet. The existence of water means a signal of bacteria that could lead to food poisoning.
They collect and graph data to create a mathematical model that represents the cooling rate, and use an exponential decay regression to determine how long a person should wait to drink the cup of hot chocolate at an optimal temperature. In Part 2, students investigate the specific heat capacity of the hot chocolate.
What should I do with my hot chocolate?
In Part 2, students investigate the specific heat capacity of the hot chocolate. They determine how much energy is needed to heat the hot chocolate to an optimal temperature after it has cooled to room temperature. Two activity-guiding worksheets are included. This engineering curriculum aligns to Next Generation Science Standards ( NGSS ).