What solvents can you use to extract THC?
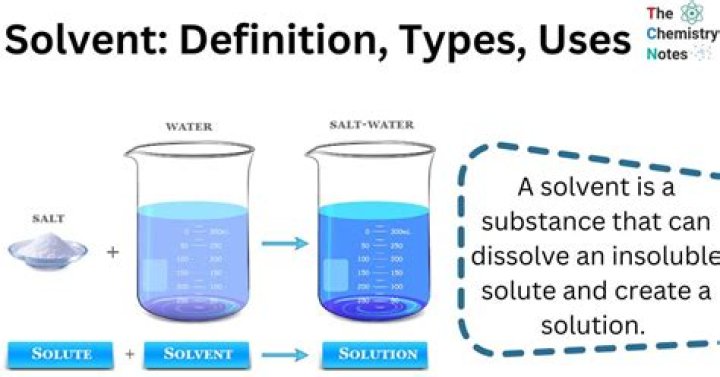
What solvents can you use to extract THC?
Currently, the most popular solvents for cannabis extractions are butane and propane, although CO2, ethanol, and heat and pressure are also employed.
Why is acetone used for extraction?
Acetone extraction might be one of several steps in the extraction and purification of useful compounds from raw materials, with other solvents being used at different stages. Acetone is used in laboratories because of its ability to dissolve many organic compounds, including oils, fats and plastics.
What solvents can be used to make RSO?
The most common solvent used to produce RSO is grain alcohol, but some other solvents like ethanol or butane are sometimes used. Flower (bud) material is placed in a large container and alcohol is added. The entire mixture is stirred and crushed into the alcohol.
What are some commonly used solvent for extraction?
Commonly used solvents like ethyl acetate (8.1 %), diethyl ether (6.9 %), dichloromethane (1.3 %) and chloroform (0.8 %) dissolved up to 10 % in water. Water also dissolves in organic solvents: ethyl acetate (3 %), diethyl ether (1.4 %), dichloromethane (0.25 %) and chloroform (0.056 %).
What is the best extraction solvent?
Methanol was identified as the most effective solvent for the extraction, resulting in the highest extraction yield (33.2%) as well as the highest content of phenolic (13.36 mg GAE/g DW), flavonoid (1.92 mg QE/g DW), alkaloid (1.40 mg AE/g DW), and terpenoids (1.25%, w/w).
How does acetone extract fat?
Since it is non-polar it can only dissolve other non-polar molecules. Fat (lipids) are non-polar. That means fats dissolve in acetone.
Is acetone a better solvent than water?
Because acetone contains non-polar methyl groups, it has the ability to interact with non-polar substances such as certain organic compounds; but because it has a polar carbonyl group, it works well with water, too.
How do you Decarb RSO after an extraction?
How to Decarboxylate
- Preheat the oven to 260°F.
- Put the extracted, winterized oil into a dry, properly sanitized tray that has been lined with parchment paper and cover with aluminum foil.
- Once the oven has heated, place the dish inside the oven.
- Inspect the material every 10 minutes for at least one hour.
Can you use denatured alcohol to make Rick Simpson oil?
Rick Simpson advocates the use of toxic solvents such as Naphtha and denatured alcohols (toxins have been added to prevent consumption) like isopropyl alcohol. High Proof Food Grade Alcohol is the most common and best choice for Cannabis Oil Extraction.
Is acetone soluble in water?
Acetone is a manufactured chemical that is also found naturally in the environment. It is a colorless liquid with a distinct smell and taste. It evaporates easily, is flammable, and dissolves in water. It is also called dimethyl ketone, 2-propanone, and beta-ketopropane.
Can acetone extract oil?
The main fatty acids observed in all extracted oil samples were linoleic, oleic and palmitic acids. Linoleic content of safflower oil extracted by hot extraction system was found between 48.23% (acetone) and 49.62% (hexane), while that oil extracted by cold method range from 48.07 (hexane) to 49.09% (acetone).