What is the structure of phenyl cation?
What is the structure of phenyl cation?
Phenyl cation
| PubChem CID | 5150536 |
|---|---|
| Structure | Find Similar Structures |
| Molecular Formula | C6H5+ |
| Synonyms | Phenyl cation |
| Molecular Weight | 77.10 |
What is the hybridization of phenyl carbocation?
The phenyl cation is also sp2 hybridized. But, empty orbital which carries + charge here is not a p orbital but sp2 orbital(33% s character). This makes it highly unstable.
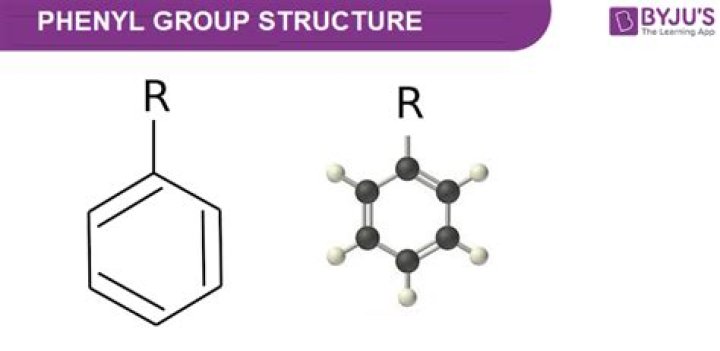
What is the structure of phenyl benzene?
Phenyl groups have six carbon atoms in a hexagonal planar structure, of which five are bonded to hydrogen atoms. They are chemically aromatic and have equal bond lengths between carbon atoms in the ring. It is poorly soluble in water….Phenyl/ Phenyle.
| Benzene (C6H6) | Phenyle (C6H5) |
|---|---|
| Six Hydrogen atoms. | Five hydrogen atoms. |
Why phenyl carbocation is not stable?
The benzene C−H bonds are sp2 hybridized. Because the electrons are closer to the nucleus with a high s character, we must use more energy to remove them and break the bond. Because of the high bond energy of the aromatic C−H bond, the phenyl carbocation is unstable.
What is the formula for phenyl?
C6H6O
Phenol/Formula
What is self ionisation of phenyl cation?
Answer: In haloarenes, the phenyl cation formed is by self ionisation,and is not stabilised by resonance. Because sp2 hybridised orbital of carbon bearing positive charge is perpendicular to the p-orbitals of the phenyl ring. Hence substitution by formation of cation (intermediate) is not possible.
Why are Aryls unstable?
The instability derives from the inability of that p orbital to overlap with the the sp2 orbitals of the carbon on the other end of the double bond. The bond angles of that carbon are too large (120*) and their highly electronegative nature prevent stabilization of the cationic center.
What is the structure of phenyl chloride?
Phenyl chloride carbonate | C7H7ClO3 – PubChem.
How do you draw the structure of a phenyl group?
Starts here4:25Phenyl group – YouTubeYouTube
Is phenyl carbo cation more stable?
Phenyl cation is more stable than cyclohexyl cation cation while phenyl anion is less stable than cyclohexyl anion.
What is phenyl composed of?
Phenyl is an emulsion of light creosote oil and water with soap. Creosote oil contains carbolic acid, creosol and other homologues of phenol which exert the necessary germicidal powder adequate incorporation of creosote oil.