What is the point-group of pf5?
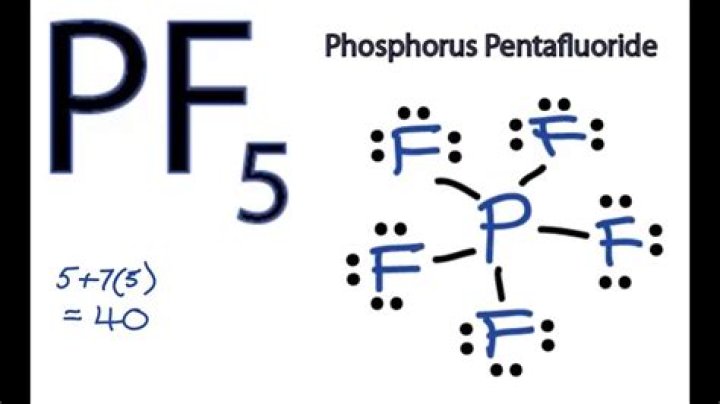
What is the point-group of pf5?
The PF5 molecule belongs to the D3h point group.
What is the symmetry of PCl5?
For example, since the PCl5 molecule has been determined to have D3h symmetry, the chart tells us that it has trigonal bipyramidal geometry using VSEPR and Lewis dot structure notation.
What is point-group PCl3?
PCl3 is trigonal pyramidal (use VSEPR theory) and so possesses the same symmetry elements as NH3 in worked example 3.2. These are E, C3 and 3 v. BCl3 is trigonal planar (use VSEPR) and possesses all the above symmetry elements: In addition, BCl3 contains a h plane and three C2 axes (see Figure 3.2).
What is the 3d structure of PCl5?
While 3d orbitals have a similar or comparable amount of energy, the energy difference between 4s and 3p orbitals means that 3d, 3p, and 4s orbitals cannot participate in the hybridization. The shape of the PCl5 molecule is Trigonal bipyramidal.
What is the point group of b2h6?
Diborane belongs to the D2h Point group and contains;Three C2 rotation axis along with 3σ planes of symmetry.
What is the point group of CH2Cl2?
group D2 2
CH2Cl2 1)belongs to point group D2 2)has one plane of symmetry, \sigma h 3)has one C2 axis of rotation | Study.com.
What is Cs point group?
In the Schoenflies notation Cn is a point group which contains the symmetry operations Cn, Cn2, and so on. Recall, from Chapter 1, that in the Schoenflies notation the reflection σh acts perpendicularly to the rotation axis while σv contains it (h, horizontal and v, vertical.)
What is b2h6 point group?
What is the point group of if7?
Structure of iodine heptafluoride, an example of a molecule with the pentagonal-bipyramidal coordination geometry….
| Pentagonal bipyramidal molecular geometry | |
|---|---|
| Examples | IF7, ZrF73− |
| Point group | D5h |
| Coordination number | 7 |
| Bond angle(s) | 90°, 72° |
What is the point group of HCN?
Point Group is Td. (d) HCN: Hydrogen cyanide is linear, so it belongs to either D∞h or C∞v. Since it does not possess an i it is C∞v .