The osmolarity of normal saline, 9 grams NaCl dissolved in water to a total volume of one liter, is a close approximation to the osmolarity of NaCl in blood (about 290 mOsm/L). Thus, normal saline is almost isotonic to blood plasma..
Besides, is blood hypertonic or hypotonic?
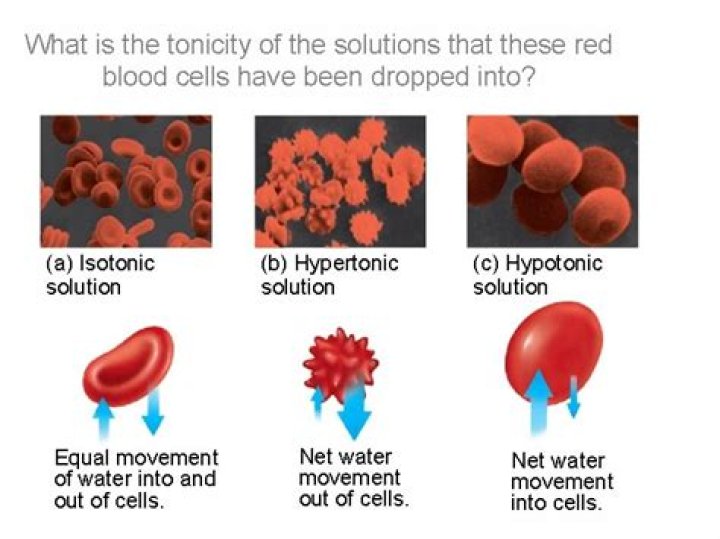
If placed in a hypotonic solution, a red blood cell will bloat up and may explode, while in a hypertonic solution, it will shrivel—making the cytoplasm dense and its contents concentrated—and may die. In the case of a plant cell, however, a hypotonic extracellular solution is actually ideal.
One may also ask, is the human body isotonic hypertonic or hypotonic? Physiology of Body Fluids Solutions that do not change the volume of a cell are said to be isotonic. A hypotonic solution causes a cell to swell, whereas a hypertonic solution causes a cell to shrink.
One may also ask, how is tonicity measured?
Tonicity is a measure of the relative concentration of non-penetrating solute on either side of a membrane. It uses the same units as molarity or osmolarity, but unlike these other measurements includes only non-penetrating solutes in the calculation. Determine the number of moles of solute.
What is the tonicity relationship between distilled water and normal blood cells?
Distilled water is water without any ions dissolved in it. We call this a hypotonic fluid and has a very low osmotic pressure. Red blood cells (or any cell for that matter) have ions, such as sodium, potassium, and other compounds dissolved in them and will have a higher osmotic pressure.
Related Question Answers
What is a hypotonic solution?
A hypotonic solution has a lower concentration of solutes than another solution. In biology, a solution outside of a cell is called hypotonic if it has a lower concentration of solutes relative to the cytosol. Due to osmotic pressure, water diffuses into the cell, and the cell often appears turgid, or bloated.What is an example of hypotonic solution?
A common example of a hypotonic solution is 0.45% normal saline (half normal saline). Common examples of hypertonic solutions are D5 in 0.9% normal saline and D5 in lactated ringers.What is an example of hypertonic solution?
Glucose is the sugar present in an individual's blood. An example of a hypertonic solution that has glucose dissolved into it is any plain glucose solution with a concentration higher than 5%. One such example is a solution of 10% dextrose in water, also known by its name D10W. This is often used as an IV fluid.What is hypertonic and hypotonic solution?
In your body, these solutes are ions like sodium and potassium. A hypotonic solution is one in which the concentration of solutes is greater inside the cell than outside of it, and a hypertonic solution is one where the concentration of solutes is greater outside the cell than inside it.How do you remember hypertonic and hypotonic?
How should I remember hypotonic vs. hypertonic= water will move out the cell, because the amount of "stuff" outside the cell is bigger. hypotonic=water will move into the cell, because the amount of "stuff" inside the cell is bigger.What happens during hypotonic?
The plant wilts because there is a loss of turgor pressure. What happens when a plant cell is placed in a hypotonic solution? The greatest concentration of water is outside the cell. Therefore, water enters the cell and fills the central vacuole, causing the contents of the plant cell to press against the cell wall.Is 10 NaCl hypertonic or hypotonic to red blood cells?
A 10 percent NaCl (sodium chloride) is hypertonic to red blood cells, as it is dependent on tonicity, which can be regarded as the relative concentration or osmotic pressure gradient of two semipermeable membrane separated solution.When would you use hypertonic or hypotonic solutions?
You want to give your patients a solution that has the tonicity that is opposite their problem most of the time. For example, if your patient is dehydrated their blood is hypertonic. They will need a hypotonic solution to bring their tonicity back within normal ranges.Is sucrose hypertonic or hypotonic?
The sucrose solution is hypertonic to the water – it is a more concentrated solution. There is a net movement of water molecules, by osmosis , from the water outside to the sucrose solution inside the Visking tubing. This makes the liquid level in the capillary tube rise.What happens to red blood cells placed in 0.9 NaCl solution?
The erythrocyte shrinks in hypertonic solutions and swells in hypotonic solutions. The red blood cell has its normal volume in isotonic NaCl. Erythrocytes remain intact in NaCl 0.9%, resulting in an opaque suspension. Distilled water on the other hand is hypotonic to red blood cells.What is the difference between isotonic hypertonic and hypotonic?
Hypotonic solution has lower solvent concentration (less denser) than the other solution. Hypertonic solution has more solvent concentration (more denser) than the other solution. Whereas Isotonic solution has similar concentrations on both sides of the semi-permeable membrane.What is the isotonic concentration of NaCl to the RBC?
A 0.9% NaCl solution is said to be isotonic: when blood cells reside in such a medium, the intracellular and extracellular fluids are in osmotic equilibrium across the cell membrane, and there is no net influx or efflux of water.What are the 3 types of osmosis?
The three types of osmotic conditions that affect living cells are called hypertonic, hypotonic, and isotonic states. These terms describe the osmotic state of the solution that surrounds a cell, not the solution inside the cell. Hypertonic conditions cause water to diffuse out of the cell, making the cell shrivel.What is a isotonic solution?
An isotonic solution refers to two solutions having the same osmotic pressure across a semipermeable membrane. This state allows for the free movement of water across the membrane without changing the concentration of solutes on either side.How can a hypotonic solution cause a cell to rupture?
A hypotonic solution means the environment outside of the cell has a lower concentration of dissolved material than the inside of the cell. If a cell is placed in a hypotonic solution, water will move into the cell. This causes the cell to swell, and it may even burst. This can cause a cell to shrink and shrivel.What does distilled water do to blood cells?
In distilled water, the concentration of water is higher outside the RBC than on the inside, as RBC contains salts and proteins. As there is difference in the osmotic pressure, the distilled water will flow into the RBC, thereby increasing its size or swell and eventually, causing it to lyse.Why is osmosis important to the human body?
The biological importance of osmosis is that it facilitates the distribution of essential nutrients in the body and the excretion of metabolic waste products. Cells have semipermeable membranes, and osmosis makes it possible for liquid solvents to pass through these cell membranes.What would happen to red blood cells in distilled water?
If RBCs are placed in distilled water, they will swell and burst due to influx of water, since ICF of RBCs is hypertonic in relation to ECF, i.e. distilled water. All animal cells, and all other cells that do not have cell walls will absorb water and eventually burst, when placed in distilled water.