What is the N 1 rule in NMR?
What is the N 1 rule in NMR?
The (n+1) Rule, an empirical rule used to predict the multiplicity and, in conjunction with Pascal’s triangle, splitting pattern of peaks in 1H and 13C NMR spectra, states that if a given nucleus is coupled (see spin coupling) to n number of nuclei that are equivalent (see equivalent ligands), the multiplicity of the …
Does first order splitting follow N 1 rule?
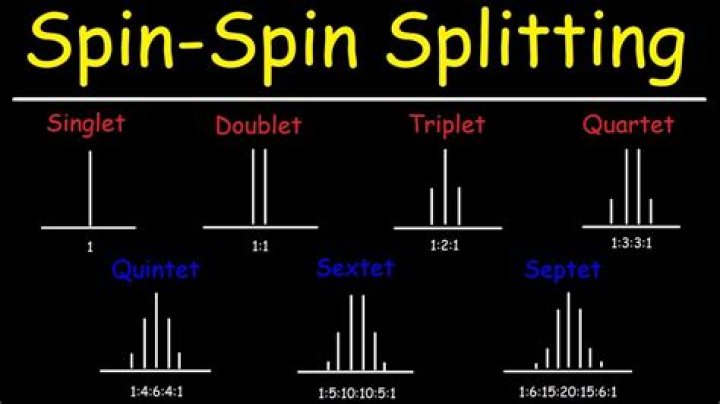
n+1 rule: When splitting is first-order, the NMR signal for a nucleus having n neighbors is split into n+1 lines. The 1H-NMR spectrum of 2-methoxybutane, illustrating the n+1 rule.
How do you calculate splitting in NMR?
To find the NMR splitting pattern, for a given hydrogen atom, count how many identical hydrogen atoms are adjacent, and then add one to that number. For example, in CH2ClCH3 below, the red hydrogen atoms are adjacent to three identical hydrogen atoms (marked in blue).
When can NMR signals be split?
Only nonequivalent protons split the signal of the given proton(s). One adjacent proton splits an NMR signal into a doublet and two adjacent protons split the signal into a triplet. For example: Ha and Hb are nonequivalent protons so they split each other’s NMR signals.
What is the splitting pattern in NMR?
In general, an NMR resonance will be split into N + 1 peaks where N = number of hydrogens on the adjacent atom or atoms. Two hydrogens on the adjacent atoms will split the resonance into three peaks with an area in the ratio of 1:2:1, a triplet.
Why does splitting occur in 1h NMR spectroscopy?
The split peaks (multiplets) arise because the magnetic field experienced by the protons of one group is influenced by the spin arrangements of the protons in an adjacent group.
What is a splitting pattern in NMR?
NMR provides information on how many hydrogen neighbors exist for a particular hydrogen or group of equivalent hydrogens. Two hydrogens on the adjacent atoms will split the resonance into three peaks with an area in the ratio of 1:2:1, a triplet. …
What is the splitting pattern in the proton NMR 1h NMR of the two methyl groups of 2 Bromopropane?
This resonance is split into a doublet by the methyl protons (n+1 = 2). This resonance is split into a 1:6:15:20:15:6:1 septet by the methyl protons.
What splitting pattern is observed in the 1h NMR spectrum for the indicated hydrogens?
The proton NMR (nuclear magnetic resonance) spectroscopy identifies the number of hydrogens in a compound which is present in a different environment. The splitting pattern follows n+1 rule, where n signifies here the number of adjacent protons.
What split ratio?
A stock split is when a company divides the existing shares of its stock into multiple new shares to boost the stock’s liquidity. The most common split ratios are 2-for-1 or 3-for-1, which means that the stockholder will have two or three shares, respectively, for every share held earlier.