What is the molecular geometry for a central atom with 3 lone pairs and 2 bonding pairs?
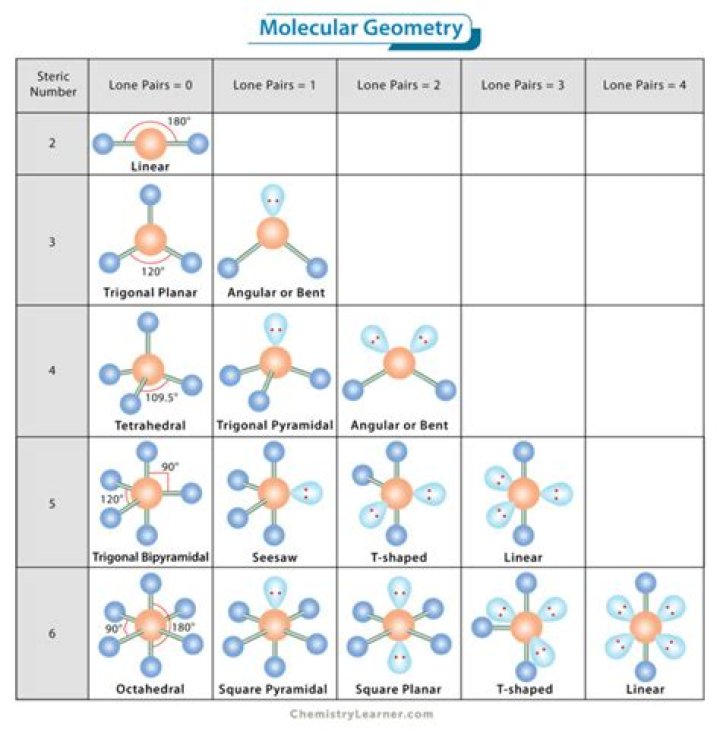
What is the molecular geometry for a central atom with 3 lone pairs and 2 bonding pairs?
| # of bonding groups/domains on ‘central’ atom | # of lone pair electrons on ‘central’ atom | Molecular Geometry |
|---|---|---|
| 3 | 0 | trigonal planar |
| 2 | 1 | bent |
| 4 | 0 | tetrahedral |
| 3 | 1 | trigonal pyramidal |
Does carbon have 3 lone pairs?
Carbon tends to form 4 bonds and have no lone pairs. Nitrogen tends to form three bonds and have on e lone pair. Oxygen tends to form two bonds and have two lone pairs.
What is the molecular geometry of a molecule with 2 outer atoms and 3 lone pairs on the central atom?
| # of bonding pair/s of electron on ‘central’ atom | # of lone pair of electrons on ‘central’ atom | Molecular Geometry |
|---|---|---|
| 2 | 1 | bent |
| 4 | 0 | tetrahedral |
| 3 | 1 | trigonal pyramidal |
| 2 | 2 | bent |
What will be the shape of molecule whose central atom is associated with 3 bonds and one lone pair?
pyramidal
So the structure is a bit distorted. Hence, the shape of a molecule which has 3 bond pairs and 1 lone pair of electrons is pyramidal.
What is the shape of a molecule with 2 bonding pairs and 2 lone pairs around the central atom?
If there are two bond pairs and two lone pairs of electrons the molecular geometry is angular or bent (e.g. H2O). Five electron pairs give a starting point that is a trigonal bipyramidal structure.
What is the molecular geometry if you have 3 single bonds and 1 lone pair?
The trigonal pyramidal geometry exists when there are 3 bonds and 1 lone pair. This is seen in ammonia (right). The bond angle for trigonal pyramidal geometries is less than 109.5∘ due to the additional repulsion from the lone pair.
Can carbon 3 bonds?
Carbon can form single, double, or even triple bonds with other carbon atoms. In a single bond, two carbon atoms share one pair of electrons. In a double bond, they share two pairs of electrons, and in a triple bond they share three pairs of electrons.
What is the lone pair of carbon?
The central atom of this molecule is carbon. Hence $2$ lone pairs are present at each Oxygen and the molecule contains two oxygen. Carbon contains $4$ valence electrons which are making double bonds with each Oxygen. So Carbon contains no lone pairs on it.
What is the molecular geometry of a molecule with 4 outer atoms and 2 lone pair on the central atom enter the molecular geometry of the molecule?
Thus, the molecular geometry of the molecule with 4 outer atoms and 2 lone pairs in the central atom is square planar.
How many outer atoms and lone pairs are present in a molecule with a trigonal pyramidal shape outer atoms lone pair’s ):?
outer atoms
A trigonal planar geometry is observed when the central atom has three outer atoms and one lone pair.
How many lone pairs are in CO2?
two lone pairs
Explanation: In the CO2 molecule, each oxygen atom has two lone pairs of electrons. The carbon atom has no lone pairs.