What is the difference between elements and compounds and mixtures?
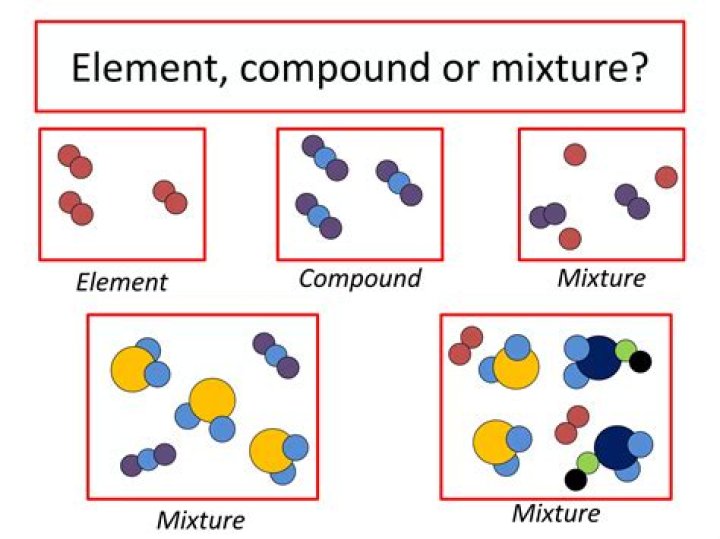
What is the difference between elements and compounds and mixtures?
Element: Pure substance consisting of one type of atom. Compound: Pure substance consisting of two or more different atoms. Mixture: Two or more different substances not chemically combined. Compound formula: Represents the combination of two or more elements in fixed proportions.
What is the difference between elements and compounds ks3?
Element: Each of more than one hundred substances that cannot be chemically interconverted or broken down into simpler substances and are primary constituents of matter. Compound: A thing that is composed of two or more separate elements.
What are 5 differences between elements and compounds?
Difference between Elements and Compounds
| ELEMENTS | COMPOUNDS |
|---|---|
| Elements can be categorized as metals, metalloids, and nonmetals. | Compounds are divided into three categories: Metallic, ionic, and molecular. |
| Elements are the single atoms. Hence they cannot be broken down. | Compounds can be broken down using chemical reactions. |
What are 3 differences between compounds and mixtures?
Difference between Compound and Mixture
| COMPOUNDS | MIXTURES |
|---|---|
| Compounds are basically of three kinds : Ionic, metallic and covalent. | Mixtures are divided into two categories : Heterogeneous and homogeneous. |
| They are pure substances. | They are impure substances. |
What are 2 differences between elements and compounds?
The difference between an element and a compound is that an element is a substance made of same type of atoms, whereas a compound is made of different elements in definite proportions. Examples of elements include iron, copper, hydrogen and oxygen.
What are mixtures compounds and elements?
Review: An element contains just one type of atom. A compound contains two or more different atoms joined together. A mixture contains two or more different substances that are only physically joined together, not chemically. A mixture can contain both elements and compounds.
What is a compound mixture and element example?
Elements are substances which are made up of only one type of atom. Compounds. A compound is formed when two or more atoms of different elements combine together chemically. An example of a compound is table-salt. It is a compound because it is made up of two different types of atom – in this case sodium and chlorine.
What is difference between elements and compounds?
An element is a material that consists of a single type of atom. Each atom type contains the same number of protons. Chemical bonds link elements together to form more complex molecules called compounds. A compound consists of two or more types of elements held together by covalent or ionic bonds.
What are 3 differences between elements and compounds?
Elements contain only one type of atom. Each atom has the same atomic number i.e., the same number of protons in their nucleus. A compound can be separated into simpler substances by chemical methods/reactions. Elements cannot be broken down into simpler substances by chemical reactions.
What is the difference between element and mixture?
Difference between element and mixture….
| ELEMENTS | MIXTURE |
|---|---|
| Elements are made up of one kind of atoms. | Mixtures are made up of two or more kinds of Compounds . |
| Elements cannot be broken down into simpler substances by any physical or chemical method. | The various constituents are seperated by simple physical means. |
What are 4 differences between compounds and mixtures?
The chemical composition of compounds is always fixed. A mixture can have a variable composition of the substances forming it. Mixtures can either be homogeneous or heterogeneous in nature. The constituents of a compound can only be separated by either chemical or electrochemical methods (like extraction).