What is the charge for formaldehyde?
What is the charge for formaldehyde?
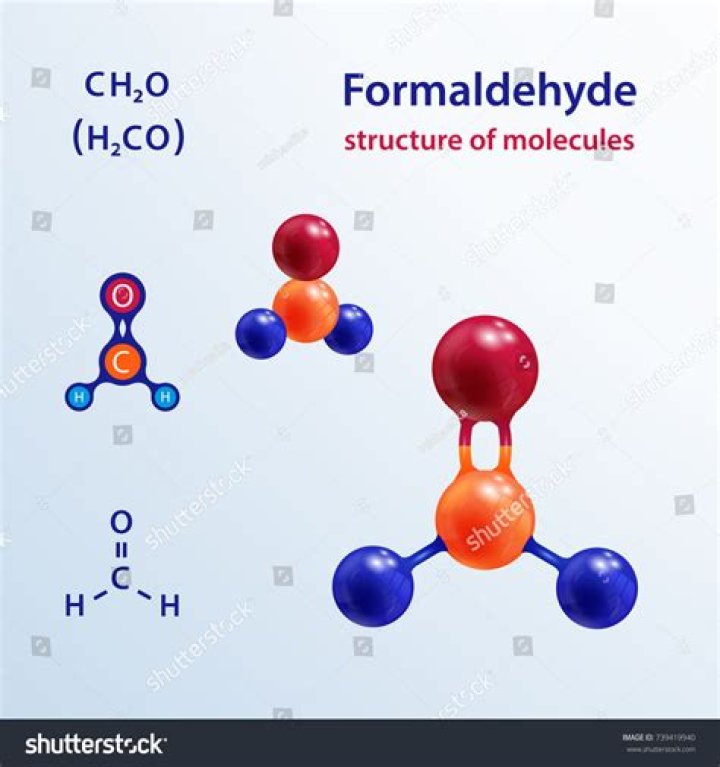
Look at the structure of formaldehyde. Oxygen has a normal valence of two, and it has two bonds in formaldehyde, so there is no formal charge on the oxygen. Carbon has a normal valence of four, and it has four bonds here.
How do you calculate formal charge from formal charge?
Since a chemical bond has two electrons, the “number of bonding electrons divided by 2” is by definition equal to the number of bonds surrounding the atom. So we can instead use this shortcut formula: Formal Charge = [# of valence electrons on atom] – [non-bonded electrons + number of bonds].
What is the formal charge of oxygen in CH2O?
1.In formaldehyde, CH2O, where carbon is the central atom, the formal charge on the oxygen is zero and the hybridization of the oxygen atom is sp2.
What is the formal charge of CoCl2?
If the molecule is an ion, add or subtract one or more electrons overall to account for the final charge. For CoCl2 (Phosgene gas): C = 4; O = 6; Cl = 7. The molecule is not ionized and has a neutral charge. Therefore, the total amount of valence electrons is 4 + 6 + (7×2) = 24.
What is the formal charge of CH3?
The formal charge of C in :CH3 is negative 1.
How do you calculate formal charge and resonance?
Calculating Formal Charge Another way of saying this is that formal charge results when we take the number of valence electrons of a neutral atom, subtract the nonbonding electrons, and then subtract the number of bonds connected to that atom in the Lewis structure.
What is the C O bond order in formaldehyde CH2O )?
Therefore, we have a bond order of two. Kathleen P.
What is the formal charge of SO2?
The other two structures In every structure, the formal charge on SO2 is zero.
What is the formal charge of ozone?
0
– The formal charge on oxygen atom 3 is ‘-1’. – Therefore the formal charge of ozone is ‘0’. Note: The formal charge of a molecule is equal to the sum of the formal charge of all the individual atoms present in the given molecule.
What is the formal charge on the oxygen atom in Co use the periodic table as needed?
Thus, the formal charge of O in CO is +1.