What is the boiling point of hydrocarbons?
What is the boiling point of hydrocarbons?
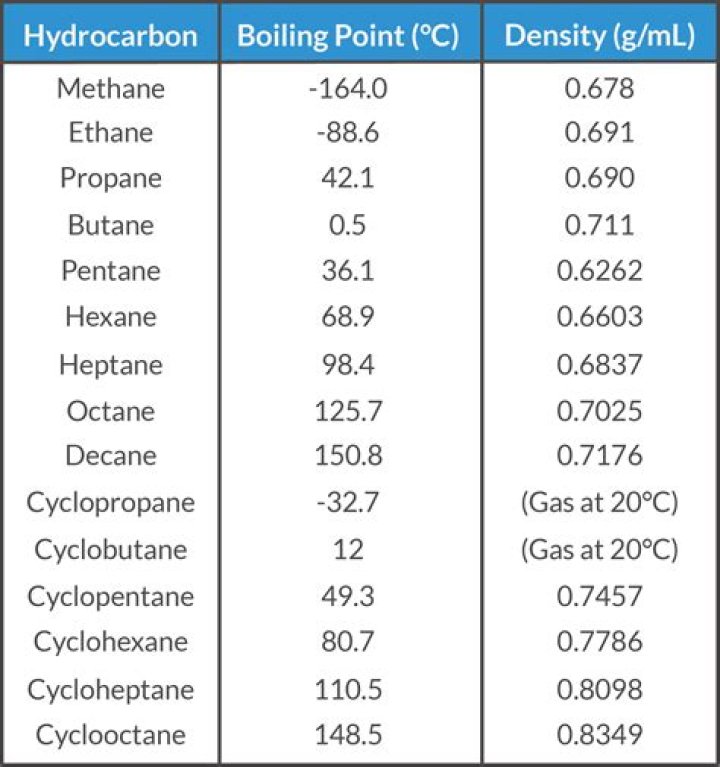
Cycloalkanes have boiling points that are approximately 20 K higher than the corresponding straight chain alkane….Boiling Points.
| Molecular Name | pentane |
|---|---|
| Formula | C 5H 12 |
| Melting Point (°C) | –130 |
| Boiling Point (°C) | 36 |
| Density (20°C)* | 0.626 g/mL |
What is boiling point in organic chemistry?
The boiling point of organic compounds can give important information about their physical properties and structural characteristics. Boiling point helps identify and characterise a compound. A liquid boils when its vapour pressure is equal to the atmospheric pressure.
What determines boiling point of organic compound?
The key thing to consider here is that boiling points reflect the strength of forces between molecules. The influence of each of these attractive forces will depend on the functional groups present. Boiling points increase as the number of carbons is increased. Branching decreases boiling point.
Why do cyclic hydrocarbons have higher boiling points?
This means cycloalkane molecules are closer on average in the liquid, and as van der Waals forces are attractive (at intermolecular separations) and their strength varies inversely with distance, the intermolecular attractions are stronger in cycloalkanes, and so the boiling point of the cycloalkanes should be higher.
What determines boiling point?
The boiling point of a liquid varies according to the applied pressure; the normal boiling point is the temperature at which the vapour pressure is equal to the standard sea-level atmospheric pressure (760 mm [29.92 inches] of mercury). At sea level, water boils at 100° C (212° F).
How do you determine boiling point?
The Formula for Boiling Point It are often calculated as: Kb = RTb2M/ΔHv, R is that the universal gas constant. Tb is that the boiling temperature of the pure solvent [in K] M is that the molar mass of the solvent.
What’s the difference between boiling point and normal boiling point?
A. “Normal boiling point” is a boiling point of a liquid at normal (standard) pressure and temperature, while “boiling point” is measured at any other conditions of pressure and temperature. “Normal boiling point” refers to 1 g of substance: “boiling point” refers to any other amount of substance.
What determines boiling point of an element?
Attractive forces between atoms in a substance affect its boiling point. The stronger the force, the harder it is to “pry” atoms apart from each other and turn them into a gas; elements with high boiling points require more heat energy to separate the atoms.
Do cyclic molecules have higher boiling points?
Cycloalkanes are types of alkanes that have one or more rings of carbon atoms in their structure. The physical properties of cycloalkanes are similar to those of alkanes, but they have higher boiling points, melting points and higher densities due to the greater number of London forces that they contain.
Why do aromatic compounds have higher boiling point?
In benzene, the only attractions between the neighbouing molecules are the van der Waals dispersion forces. There is no permanent dipole on the molecule. The higher boiling point is presumably due to the ease with which temporary dipoles can be set up involving the delocalized electrons. Methylbenzene boils at 111°C.
What is boiling point Class 12?
Boiling point is the temperature of a liquid at which its vapour pressure becomes equal to the atmospheric pressure.