What is the acid dissociation constant for acetic acid?
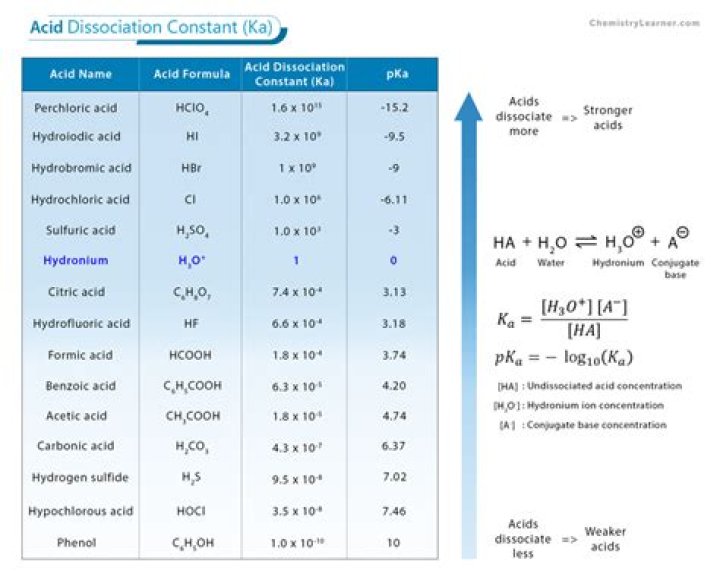
What is the acid dissociation constant for acetic acid?
E1: Acid Dissociation Constants at 25°C
| Name | Formula | K a1 |
|---|---|---|
| Acetic acid | CH3CO2H | 1.75 × 10−5 |
| Arsenic acid | H3AsO4 | 5.5 × 10−3 |
| Benzoic acid | C6H5CO2H | 6.25 × 10−5 |
| Boric acid | H3BO3 | 5.4 × 10−10* |
Is dihydrogen phosphate a weak acid?
Strong acids are 100% ionized in solution. Weak acids are only slightly ionized. Phosphoric acid is stronger than acetic acid and so is ionized to a greater extent….Strong and Weak Acids and Acid Ionization Constant.
| Acid | Conjugate Base |
|---|---|
| Weak Acids | |
| H3PO4 (phosphoric acid) | H2PO4− (dihydrogen phosphate ion) |
| CH3COOH (acetic acid) | CH3COO− (acetate ion) |
What is the conjugate acid of dihydrogen phosphate?
Phosphoric Acid
Dihydrogen phosphate
| Names | |
|---|---|
| Chemical formula | H2O4P−1 |
| Molar mass | 96.986 g·mol−1 |
| Conjugate acid | Phosphoric Acid |
| Related compounds |
How do you find the dissociation constant of acetic acid?
Acetic acid is a weak acid with an acid dissociation constant Ka=1.8×10−5 K a = 1.8 × 10 − 5 .
Is dissociation of acetic acid endothermic?
As expected for an endothermic reaction, the reaction’s equilibrium constant increases at higher temperatures; thus, acetic acid is a stronger weak acid at higher temperatures than it is at lower temperatures.
Is acetic acid a weak acid?
Acetic acid (found in vinegar) is a very common weak acid. Its ionization is shown below. The ionization of acetic acid is incomplete, and so the equation is shown with a double arrow. The extent of ionization of weak acids varies, but is generally less than 10%.
Is chloric acid a strong acid?
Chloric acid, HClO3, is an oxoacid of chlorine, and the formal precursor of chlorate salts. It is a strong acid (pKa ≈ −2.7) and oxidizing agent.
Is H3PO4 a conjugate acid?
Table of acids and their conjugate bases
| Acid | Conjugate base |
|---|---|
| H3PO4 Phosphoric acid | H2PO − 4 Dihydrogen phosphate ion |
| CH3COOH Acetic acid | CH3COO− Acetate ion |
| HF Hydrofluoric acid | F− Fluoride ion |
| H2CO3 Carbonic acid | HCO − 3 Hydrogen carbonate ion |
Is PO43 an acid or base?
More on Acid and Base
| A | B |
|---|---|
| H2CO3, H2PO4-, NH4+, HCN, HCO3-, HPO42- | Very weak acids |
| HCO3-, HPO42-, NH3, CN, CO32-, PO43- | Weak bases |
| Strong Acids | Give up proton easily and are essentially 100% dissociated in water |
| Weak Acids | give up proton with difficulty and are less than 100% dissociated in water |
What is the dissociation constant for NH4OH?
1.8 × 10^-5
Dissociation constant of NH4OH is 1.8 × 10^-5 .