What is the absorption spectra of the sun?
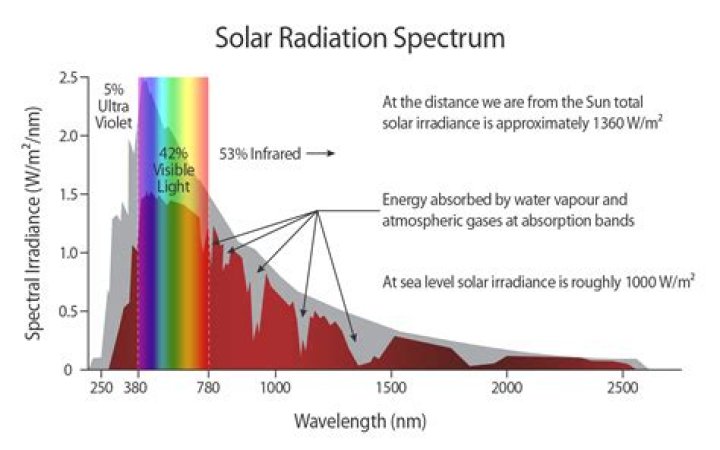
What is the absorption spectra of the sun?
The Sun produces an absorption spectrum, with dark lines across its spectrum. Chemical elements in the Sun’s corona absorb specific wavelengths of light so their electrons are excited to higher energy levels.
What kind of Spectra is the sun?
emission spectrum
The full electromagnetic spectrum. The spectrum of the Sun appears as a continuous spectrum and is frequently represented as shown below. This type of spectrum is called an emission spectrum because what you are seeing is the direct radiation emitted by the source.
What are the spectral lines of the sun?
In physics and optics, the Fraunhofer lines are a set of spectral absorption lines named after the German physicist Joseph von Fraunhofer (1787–1826). The lines were originally observed as dark features (absorption lines) in the optical spectrum of the Sun (white light) .
In what range of wavelength does the sun’s spectrum peak?
approximately 500 nanometers
The maximum wavelength output from the surface of the sun (originating from the photosphere) is approximately 500 nanometers (varying from exact measurements of 483 to 520 nm, depending on the temperature used to represent the surface of the sun, which is not clearly defined), while wavelength output from the inner …
What are characteristics of absorption spectra?
An absorption spectrum occurs when light passes through a cold, dilute gas and atoms in the gas absorb at characteristic frequencies; since the re-emitted light is unlikely to be emitted in the same direction as the absorbed photon, this gives rise to dark lines (absence of light) in the spectrum.
What is an absorption spectrum definition?
Definition of absorption spectrum : an electromagnetic spectrum in which a decrease in intensity of radiation at specific wavelengths or ranges of wavelengths characteristic of an absorbing substance is manifested especially as a pattern of dark lines or bands.
What are absorption lines in a spectrum?
An absorption line will appear in a spectrum if an absorbing material is placed between a source and the observer. This is seen in the spectra of stars, where gas (mostly hydrogen) in the outer layers of the star absorbs some of the light from the underlying thermal blackbody spectrum. …
What is the lambda max of the sun?
501.7 nm
Let’s try to calculate the temperature of the Sun’s surface: Find the peak wavelength of a solar spectrum. It’s approximately λmax = 501.7 nm (or 5.017 * 10⁻⁷ m in the scientific notation).
What is the absorption spectrum of a pigment?
Each type of pigment can be identified by the specific pattern of wavelengths it absorbs from visible light, which is the absorption spectrum. Chlorophyll a absorbs light in the blue-violet region, while chlorophyll b absorbs red-blue light.
What is difference between emission and absorption spectra?
The main difference between emission and absorption spectra is that an emission spectrum has different coloured lines in the spectrum, whereas an absorption spectrum has dark-coloured lines in the spectrum.
How do stars absorb absorption spectrum?
An absorption spectrum is produced when a continuum passes through “cooler” gas. Photons of the appropriate energies are absorbed by the atoms in the gas. The atmospheres of stars act as a cooler blanket around the hotter interior of a star so that typical stellar spectra are absorption spectra.


