What is structure of catechol?
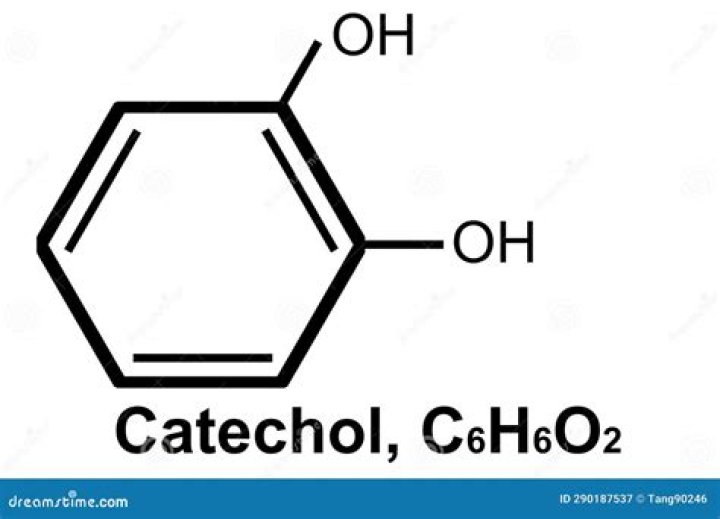
What is structure of catechol?
Catechol (/ˈkætɪtʃɒl/ or /ˈkætɪkɒl/), also known as pyrocatechol or 1,2-dihydroxybenzene, is a toxic organic compound with the molecular formula C6H4(OH)2. It is the ortho isomer of the three isomeric benzenediols. This colorless compound occurs naturally in trace amounts.
What is a catechol group?
A catechol is an unsaturated six-carbon ring (phenolic group) with two hydroxyl groups attached to adjacent carbons (dihydroxyphenol).
Which of the following structure represents catechol?
Catechol is the specific name of benzen-1,2-diol.
What is the structure of resorcinol?
C6H6O2
Resorcinol/Formula
What is the difference between catechol and Pyrocatechol?
As nouns the difference between pyrocatechol and catechol is that pyrocatechol is (organic compound) a dihydric phenol, benzene-1,2-diol, present in some tars while catechol is (organic compound) the biologically important diphenol ortho-dihydroxy benzene; isomeric with hydroquinone and resorcinol.
How do you make catechol?
Catechol can be prepared by the oxidation of salicylaldehyde1 and of phenol;2 by the demethylation of guaiacol with aluminum chloride3 and hydriodic acid;4 by hydrolysis of ortho halogen-substituted phenols or ortho dihalobenzenes under suitable conditions;5 and by the replacement of sulfonic groups by alkali fusion.
How does catechol work?
Catechol oxidase (also known as catecholase) is an enzyme present in most fruits and vegetables. It facilitates the browning of cut or bruised produce by catalyzing a reaction between the substrate molecule catechol and atmospheric oxygen (O2).
Where is catechol found?
vacuoles
Catechol is present in small quantities in the vacuoles of cells of many plant tissues. Catechol oxidase is present in the cell cytoplasm. If the plant tissues are damaged, the catechol is released and the enzyme converts the catechol to ortho-quinone, which is a natural antiseptic.
What is catechol substrate?
The substrates of catecholase are catechol and oxygen. The substrates react with one another within the active site of the enzyme. The products formed by this reaction are benzoquinone and water; since benzoquinone has a brown color, you can see that the reaction has taken place.
How do you identify resorcinol?
Resorcinol is a very white crystalline solid that becomes pink on exposure to light if not completely pure. Burns although ignition is difficult. Density approximately 1.28 g / cm3. Irritating to skin and eyes.
What does resorcinol do for the skin?
Resorcinol is used to treat acne, seborrheic dermatitis, eczema, psoriasis, and other skin disorders. It is also used to treat corns, calluses, and warts. Resorcinol works by helping to remove hard, scaly, or roughened skin.
Where is catechol used?
Catechol (1,2-dihydroxybenzene) is used in a variety of applications. It is used as a reagent for photography, dyeing fur, rubber and plastic production and in the pharmaceutical industry (Merck, 1989; Milligan and Häggblom, 1998).