What is salt water in chemistry?
What is salt water in chemistry?
CHEMISTRY GLOSSARY Salt water is the water of the sea and the ocean. This water contains a relatively high percentage of dissolved salt (about 35 g of salt per 1 000 g of sea water.). About 90 % of that salt would be sodium chloride, or ordinary table salt. The salinity of ocean water varies.
What is the formula of salt and water?
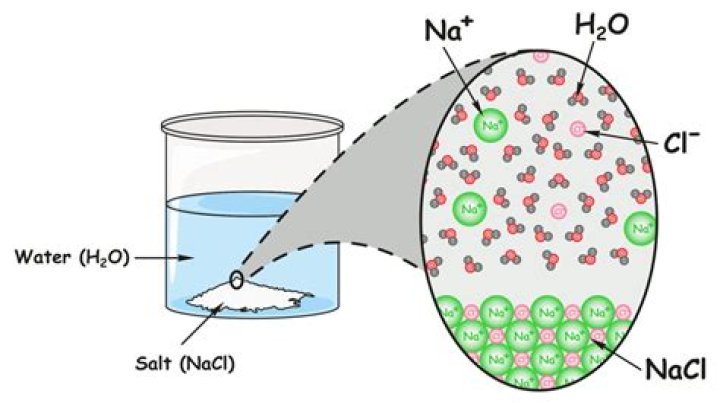
Table salt is an ionic compound, which breaks into its component ions or dissociates in water. These ions are Na+ and Cl-. The sodium and chlorine atoms are present in equal amounts (1:1 ratio), arranged to form a cubic crystal lattice. The molecular formula of table salt—sodium chloride—is NaCl.
What chemicals are in sea water?
Chemical and physical properties of seawater The six most abundant ions of seawater are chloride (Cl−), sodium (Na+), sulfate (SO24−), magnesium (Mg2+), calcium (Ca2+), and potassium (K+). By weight these ions make up about 99 percent of all sea salts.
Is sea water a substance?
Seawater is a mixture of many different substances. Water, H2O, is a pure substance, a compound made of hydrogen and oxygen. Although water is the most abundant substance on earth, it is rarely found naturally in its pure form. Most of the time, pure water has to be created.
What is sea water called?
Saline water (also called salt water, salt-water or saltwater) is water with a high amount of salt in it. It often means the water from the seas (sea water) and oceans. Almost all the water on Earth is saline.
What is the chemical name for water?
Oxidane
Water
Water/IUPAC ID
Dihydrogen monoxide is a name for the water molecule, which comprises two hydrogen atoms and one oxygen atom (H2O).
What is the seawater pH?
Today, average ocean pH is about 8.1. This might not seem like much of a difference, but the relationship between pH and acidity is not direct. Each decrease of one pH unit is a ten-fold increase in acidity.
What salts are in sea water?
The two ions that are present most often in seawater are chloride and sodium. These two make up over 90% of all dissolved ions in seawater. By the way, the concentration of salt in seawater (salinity) is about 35 parts per thousand.
What is the solvent of sea water?
In the solution of seawater, water is the solvent. The hydrogen and oxygen atoms in water molecules make up about 96.5 percent of the mass of seawater.
What solution is sea water?
Ocean water is a homogenous mixture of salt in water. It contains about 3 percent sodium chloride. Ocean water is the type of mixture called a solution, because the salt is dissolved in the water. Water is the solvent, and sodium chloride is the solute.
Is sea water is a pure substance?
Seawater is a mixture of many different substances. Some of these substances can be observed when the water in seawater evaporates and leaves behind salt. Water, H2O, is a pure substance, a compound made of hydrogen and oxygen. As water evaporates, it distills, or leaves the salt behind.