What is rate of a reaction in Igcse chemistry?
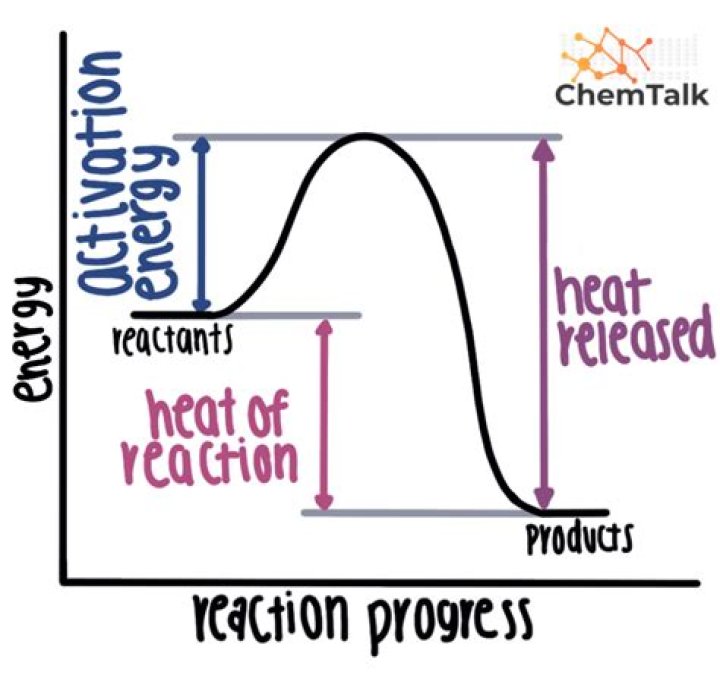
What is rate of a reaction in Igcse chemistry?
The rate of a reaction is a measure of how quickly a reactant is used up, or a product is formed.
What is the rate of reaction in chemistry?
reaction rate, in chemistry, the speed at which a chemical reaction proceeds. It is often expressed in terms of either the concentration (amount per unit volume) of a product that is formed in a unit of time or the concentration of a reactant that is consumed in a unit of time.
What is a chemical reaction Igcse?
In chemical reactions, new chemical products are formed that have very different properties to the reactants. Most chemical reactions are impossible to reverse. Energy changes also accompany chemical changes and energy can be given out (exothermic) or taken in (endothermic)
How do you calculate the rate of a chemical reaction?
For a reaction of A → B the reaction rate is (change of concentration)/time. If plotted on a graph, the concentration of A would decrease over time and the concentration of B would increase. The gradient of the graph indicates the rate of reaction – the steeper the gradient the faster the rate of reaction.
How do you find the rate of reaction?
Measuring Reaction Rates
- Reaction rate is calculated using the formula rate = Δ[C]/Δt, where Δ[C] is the change in product concentration during time period Δt.
- The rate of reaction can be observed by watching the disappearance of a reactant or the appearance of a product over time.
How do you calculate rate of reaction example?
Key Points
- Reaction rate is calculated using the formula rate = Δ[C]/Δt, where Δ[C] is the change in product concentration during time period Δt.
- The rate of reaction can be observed by watching the disappearance of a reactant or the appearance of a product over time.
What is the average rate of reaction?
The average rate of reaction is an average rate, obtained by taking the change in concentration over a time period. The rate at which chemical substances react varies greatly. Usually, a reaction rate involves the change in the concentration of a substance over a given period of time.
What is equilibrium Igcse?
Equilibrium is dynamic i.e. the molecules on the left and right of the equation are changing into each other by chemical reactions constantly and at the same rate. The concentration of reactants and products remains constant (given there is no other change to the system such as temperature and pressure)