What is peritectic reaction?
What is peritectic reaction?
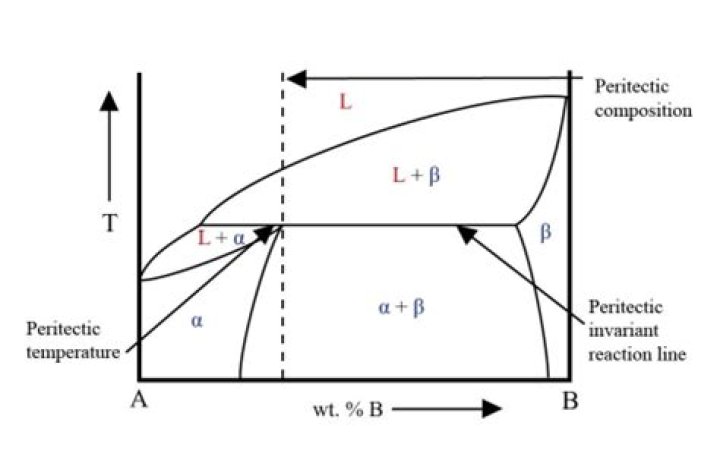
A peritectic reaction is a reaction where a solid phase and liquid phase will together form a second solid phase at a particular temperature and composition – e.g. Diagram showing the Peritectic reaction, where a liquid and solid together form a new solid phase.
What is peritectic phase diagram?
Peritectic point – The point on a phase diagram where a reaction takes place between a previously precipitated phase and the liquid to produce a new solid phase. When this point is reached, the temperature must remain constant until the reaction has run to completion. A peritectic is also an invariant point.
What is peritectic reaction example?
DEF. A peritectic reaction is a three-phase reaction by which, on cooling, two phases (one of them liquid) react to give a single new solid phase. Liquid + solid α → solid β . The composition of the β which forms (in this example) is 50 at% B.
What is the type of transformation in peritectic reaction?
During the peritectic reaction all three phases (primary, secondary, and liquid phases) are in contact with each other. In the peritectic transformation the liquid and primary phases are isolated by the secondary phase, and the transformation takes place by long-range diffusion through the secondary phase.
What is peritectic reaction in iron carbon diagram?
Peritectic Reaction • Peritectic reaction: at 0.16% C and 14930 C δ(0.11% C) + L(0.51%C) ↔ γ (0.16%C) • In peritectic reaction, the liquid and δ iron transforms into austenite (containing 0.16% C). The peritectic reaction occurs at a constant temperature. This is known as peritectic temperature and is 1493°C.
What is meant by peritectic and Peritectoid reactions?
In binary systems, the peritectoid is the transformation of two solid phases in an alloy system, forming a new phase. These reactions happen in a similar way to peritectic transformations (see Peritectic), however, they only occur in the solid state.
Which of the following are peritectic systems?
Which of the following are Peritectic systems? Explanation: Peritectic systems – Pt – Ag, Ni – Re, Fe – Ge, Sn-Sb (babbit).
What are eutectic and peritectic reactions?
> The difference between eutectic and peritectic is that eutectic is describing the thermodynamic equilibrium conditions where a liquid coexists with two solid phases while peritectic is describes the isothermal reversible reaction of a liquid phase and a solid phase to form a second solid phase during cooling.
What is the peritectic reaction at 1495 C?
1. What is the Peritectic reaction at 1495°C? L (0.53% C) + δ(0.09% C) → γ(0.17% C).
At what temperature does peritectic reaction occur?
1495°C
Peritectic temperature 1495°C (2723°F). Austenite (γ) phase (fcc) with carbon in interstitial solid solution. Because the outcome of the reaction is not shown in the final microstructure of carbon and low alloy steels, little attention is paid to the peritectic reaction in solidification stages.
What is peritectic steel?
Steels in range II are known as peritectic steels and are the most difficult to cast because they experience transformation from δ to γ that coincides with the final stage of solidification. While in range I, δ to γ transformation starts and ends in the solid state.