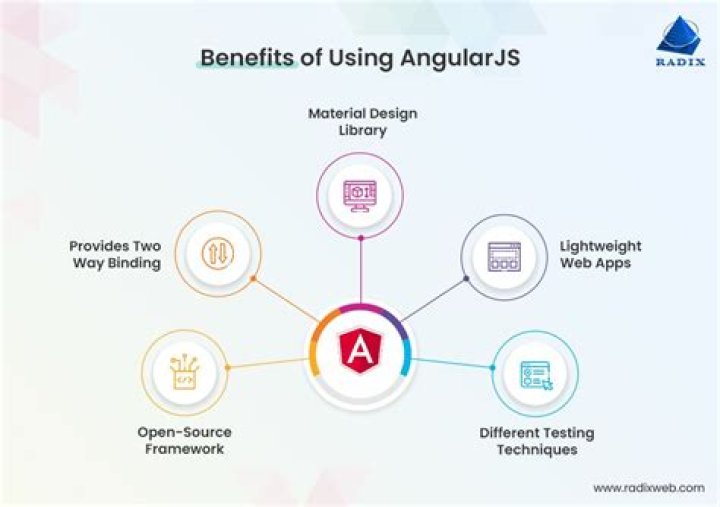
What is Meona?
What is Meona?
Meona is the perfect clinical software for all clinical fields of your hospital. From patient admission to discharge, from medication to reporting, Meona delivers intelligent solutions for your documentation and provides the desired overview.
What is the name of ch3ona?
SODIUM METHOXIDE
Sodium methoxide
| PubChem CID | 10942334 |
|---|---|
| Structure | Find Similar Structures |
| Chemical Safety | Laboratory Chemical Safety Summary (LCSS) Datasheet |
| Molecular Formula | CH3ONa or CH3NaO |
| Synonyms | Sodium methanolate SODIUM METHOXIDE Sodium methylate 124-41-4 Methoxysodium More… |
How do you make methanol from Methoxide?
To prepare Sodium Methoxyde Solution (1 M) , take 4.6 ml in dry methanol , add 2.3 gram of freshly cut sodium metal till it is slowly dissolved. Use this turbid solution as 1M sodium methoxide for the reaction.
What is SMO chemical?
Sodium Methylate (SMO)
How do you make Methoxide?
On a large scale, potassium methoxide is produced by decomposing potassium amalgam with methanol, which is produced by the chloralkali-electrolysis of potassium chloride by the amalgam process. Impurities of the resulting potassium methoxide in methanol with metallic mercury can be eliminated by ultrafiltration.
Is sodium methoxide a Alkoxide?
From reducing metals The alcohol serves as an acid, and hydrogen is produced as a by-product. A classic case is sodium methoxide produced by the addition of sodium metal to methanol: The metal hydride removes the hydrogen atom from the hydroxyl group and forms a negatively charged alkoxide ion.
What is 254 SMO material?
View in webshop. Sandvik 254 SMO is a high-alloy austenitic stainless steel developed for use in seawater and other aggressive chloride-bearing media. The steel is characterized by the following properties: Excellent resistance to pitting and crevice corrosion, PRE = ≥42.5* High resistance to general corrosion.
Is sodium methoxide a catalyst?
Sodium methoxide: a simple but highly efficient catalyst for the direct amidation of esters†
What is a methoxide catalyst?
The methoxide ion, –OCH3, is the active catalyst for the production of methyl esters. It is this chemical unit that attacks the triglyceride molecules and produces the methyl esters. Most small producers create the methoxide ions needed for the reaction by dissolving sodium hydroxide or potassium hydroxide in methanol.
Is methanol an alkoxide?
The alcohol serves as an acid, and hydrogen is produced as a by-product. A classic case is sodium methoxide produced by the addition of sodium metal to methanol: The metal hydride removes the hydrogen atom from the hydroxyl group and forms a negatively charged alkoxide ion.
Can u drink methanol?
Drinking just 25-90 mL (0.7-3.0 ounces) of methanol can be fatal without proper medical treatment. Since methanol is an industrial chemical, it should not be consumed in any quantity, just as people would normally not mix gasoline or other industrial chemicals into legitimate alcoholic beverages.