What is low grade energy?
.
In this way, what is low grade heat?
High grade heat: temperature is higher than 650 °C, medium grade heat: temperature ranges between 200-277 °C and 650 °C, low grade heat: temperature is lower than 200-277 °C. As the temperature grows the size of slices reduces.
Similarly, why is Heat called a degraded form of energy? All kinetic energy (movement) causes friction, a force which dissipates energy as heat. Thus heat could be described as the 'graveyard' - it is where all kinetic energy ends up. Thus heat is also described as a degraded form of energy.
Simply so, what are grades of energy?
There are four grades. The weight grade determines how much energy there is per pound of energy resource. For example, coal has about 12.7 thousand BTU/lb, natural gas about 10 thousand BTU/lb, oil about 19.3 thousand BTU/lb, and an electric battery typically has 100 BTU/lb.
What do you mean by thermodynamics?
Thermodynamics is the branch of physics that deals with heat and temperature, and their relation to energy, work, radiation, and properties of matter. Statistical thermodynamics, or statistical mechanics, concerns itself with statistical predictions of the collective motion of particles from their microscopic behavior.
Related Question AnswersWhy heat is a low grade energy?
Heat is low grade energy - If you have 100kJ of energy in the form of heat you CAN NOT exchange it anywhere, any time for 100kJ of energy of any other form. Technically speaking - We say heat can not be transformed into any other form of energy with 100% efficiency.Is heat a waste product?
Waste heat is heat that is produced by a machine, or other process that uses energy, as a byproduct of doing work. All such processes give off some waste heat as a fundamental result of the laws of thermodynamics.How is heat wasted?
Waste heat is the unused heat given to the surrounding environment (in the form of thermal energy) by a heat engine in a thermodynamic process in which it converts heat to useful work. Waste heat is often dissipated into the atmosphere, or large bodies of water like rivers, lakes and even the ocean.What is another term for waste heat?
Definition of waste heat. : heat rejected or escaping from furnaces of various types (as coke ovens, cement kilns, or steel furnaces) after it has served its primary purpose.Is heat a byproduct of metabolism?
Heat is both a byproduct of metabolism and a form of energy that influences the speed at which metabolism occurs, otherwise known as metabolic rate.What is a heat dump?
heat-dump. Noun. (plural heat dumps) (thermodynamics) A structure or environment into which waste heat can be transferred.What is meant by quality of energy?
Energy quality is the contrast between different forms of energy, the different trophic levels in ecological systems and the propensity of energy to convert from one form to another. The concept refers to the empirical experience of the characteristics, or qualia, of different energy forms as they flow and transform.How is wasted heat energy used in the human body?
Thermal Energy Created by the Human Body. The body is a heat engine. It converts chemical energy of the food consumed into both heat to sustain metabolism and work. The human body primarily rejects heat to the environment from the body surface by convection, radiation, or evaporation.What are the different forms of energy?
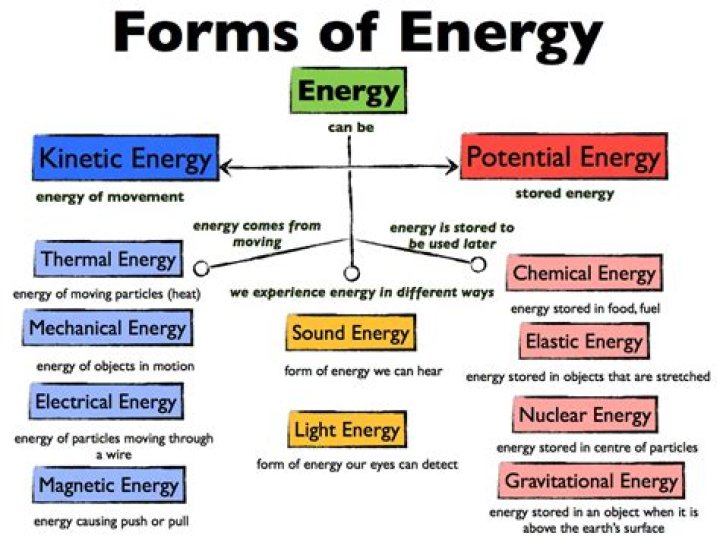
Various forms of energy such as light, heat, sound, electrical, nuclear, chemical, etc are briefly explained.- Chemical energy. Chemical energy is energy stored in the bonds of chemical compounds (atoms and molecules).
- Electrical Energy.
- Mechanical Energy.
- Thermal energy.
- Nuclear energy.
- Gravitational Energy.
- Related Resources.


