What is Km and Vmax definition
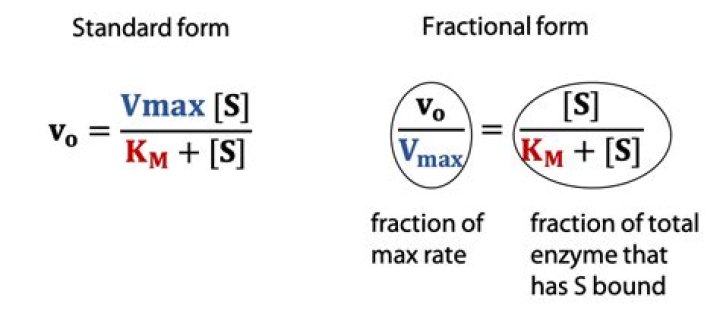
Vmax is the maximum rate of an enzyme catalysed reaction i.e. when the enzyme is saturated by the substrate. Km is measure of how easily the enzyme can be saturated by the substrate. Km and Vmax are constant for a given temperature and pH and are used to characterise enzymes.
What Vmax means?
The maximal velocity of the reaction (or maximal rate) Vmax is the rate attained when the enzyme sites are saturated with substrate, i.e. when the substrate concentration is much higher than the KM. Examples: Q8W1X2, Q9V2Z6. The Vmax value depends on environmental conditions, such as pH, temperature and ionic strength.
Is Vmax dependent on Km?
For practical purposes, Km is the concentration of substrate which permits the enzyme to achieve half Vmax. An enzyme with a high Km has a low affinity for its substrate, and requires a greater concentration of substrate to achieve Vmax.”
What is Km value in biology?
km value or Michaelis constant is defined as the substrate concentration at which half of the enzyme molecules are forming (ES) complex or concentration of the substrate when the velocity of the enzyme reaction is half the maximum value.What is the unit of Km and Vmax?
Vmax is the maximum enzyme velocity in the same units as Y. It is the velocity of the enzyme extrapolated to very high concentrations of substrate, so its value is almost always higher than any velocity measured in your experiment. Km is the Michaelis-Menten constant, in the same units as X.
What is a low Km?
Since the Michaelis-Menton constant Km is the concentration of substrate at 0.5Vmax, it is an inverse measure of its substrate affinity, because a lower Km indicates that less substrate is needed to reach a certain reaction speed. Hence, a low Km means a high substrate affinity.
What does a low Vmax indicate?
A lower Vmax means that the enzyme is operating in sub-optimal conditions.
Who gave lock and key model?
The lock and key model, originally proposed by Emil Fischer, describes interactions which are rigid in nature (Kastritis and Bonvin, 2013a; Fischer, 1894). Here both interaction interfaces are complementary in shape and negligible conformational changes take place on binding.What is meant by Vmax value Class 11?
Vmax is the maximum velocity of the reaction for the given concentration of enzyme. … This maximum velocity is Vmax.
How do you calculate KMA from Km and Vmax?Yes Kcat=Vmax/[E], where [E] = total enzyme, i.e., free enzyme and enzyme bound to substrate or intermediate. Hi Farhadi, It’s true that to calculate Kcat of an enzyme , you can use Kcat=Vmax/[Et].
Article first time published onIs Vmax ever reached?
Maximal Velocity (Vmax): Increasing the substrate concentration indefinitely does not increase the rate of an enzyme-catalyzed reaction beyond a certain point. This point is reached when there are enough substrate molecules to completely fill (saturate) the enzyme’s active sites.
What does km stand for in distance?
kilometre (km), also spelled kilometer, unit of length equal to 1,000 metres and the equivalent of 0.6214 mile (see metric system).
What can kilometers measure?
Kilometer is a unit used to measure length or distance. It is a unit used in metric system. The distance can also be measured in miles, which can be expressed in terms of kilometer.
How do you calculate kilometers?
Multiply the distance, if given in miles, by the factor 1.609 to convert to kilometers. For example, 86 miles converts to 86 x 1.609 or 138.374 kilometers. Convert the speed from miles per hour to kilometers per hour. If the speed is given in miles per hour, then multiply by 1.609.
Can Km and Vmax be negative?
Km can never be a negative number because Km denotes the concentration of an enzyme substrate at 1/2 Vmax of enzyme activity. … At a certain point the enzyme activity [V] is saturated.at high [S]. That is the Vmax. 1/2 of the Vmax is the Km.
Why does low Km means high affinity?
Km is the concentration of substrate that gets you half way to Vmax. Low Km means less substrate is necessary, meaning higher affinity.
Why do uncompetitive inhibitors decrease km?
Since uncompetitive inhibitors only block processes beyond ES formation, one might expect only Vmax to be suppressed with no effect on Km, but as the inhibitor binds to and stabilizes the ES complex, it makes it more difficult for S to dissociate or be converted to product, increasing enzyme affinity for S and so …
Is HIGH Km or low Km better?
Why is lower km better? The value of KM is inversely related to the affinity of the enzyme for its substrate. High values of KM correspond to low enzyme affinity for substrate (it takes more substrate to get to Vmax ). Low KM values for an enzyme correspond to high affinity for substrate.
Is lower or higher Km better?
Km is like a measure of fuel efficiency. The less fuel you need to reach “normal speed”, the more efficient your car is. … The less substrate they need to reach half of their maximum speed, the more efficient they are. So if the Km is low, you have a really efficient enzyme.
Why is Vmax not exceeded by any further rise in the substrate concentration?
The reaction ultimately reaches a maximum velocity which is not exceeded by any further rise in concentration of the substrate because the enzyme molecule is fewer than substrate molecules and after saturation of these molecules, there are no free enzymes to bind with the additional substrate molecules.
Why is it called lock and key theory?
The specific action of an enzyme with a single substrate can be explained using a Lock and Key analogy first postulated in 1894 by Emil Fischer. In this analogy, the lock is the enzyme and the key is the substrate. Only the correctly sized key (substrate) fits into the key hole (active site) of the lock (enzyme).
What is the induced fit theory?
allosteric control In allosteric control. …the basis of the so-called induced-fit theory, which states that the binding of a substrate or some other molecule to an enzyme causes a change in the shape of the enzyme so as to enhance or inhibit its activity.
What is substrate in bio?
In biology, a substrate is the surface on which an organism (such as a plant, fungus, or animal) lives. A substrate can include biotic or abiotic materials and animals. For example, encrusting algae that lives on a rock (its substrate) can be itself a substrate for an animal that lives on top of the algae.
What is the unit of k cat?
The units of kcat are moles of product/sec divided by moles of enzyme. In consequence, kcat resulted in 1/time units.
How do you solve k cat?
Usually it is straightforward to express this (or convert to ) moles/minute/mg of protein. If you know the concentration of enzyme sites you’ve added to the assay (Et) then you can calculate the catalytic constant Kcat. It is defined to equal Vmax/Et.
What is km Vmax and kcat?
Vmax is equal to the product of the catalyst rate constant (kcat) and the concentration of the enzyme. … Km is the concentration of substrates when the reaction reaches half of Vmax. A small Km indicates high affinity since it means the reaction can reach half of Vmax in a small number of substrate concentration.
How do I find my ki?
Thus, KI can be determined by plotting the slope values vs. [I]. The resulting secondary plot or “replot” will have a Y-axis intercept of KM/Vmax and a slope of KM/VmaxKI. The value of KI is the slope/intercept of this replot.
What is Vmax affected by?
Vmax is a rate of reaction. It will have units of: or or etc. min sec min Vmax depends on the structure the enzyme itself and the concentration of enzyme present. KM is a the concentration substrate required to approach the maximum reaction velocity – if [S]>>Km then Vo will be close to Vmax.
How does uncompetitive inhibition affect Km and Vmax?
Uncompetitive inhibitors can only bind to the ES complex. Therefore, these inhibitors decrease Km because of increased binding efficiency and decrease Vmax because they interfere with substrate binding and hamper catalysis in the ES complex.
How long is a km?
KilometerMileFeet1 km = 3280.84 ft1 mile = 5,280 ftMeters1 km = 1000 m1 mile = 1609.344 mInches1 km = 39,370 in1 mile = 63,360 in
What is 1km example?
A kilometer is a unit of length in the metric system of measurement that is equal to 1000 meters. To show how far 1 kilometer is, we can use the length of an Airbus 747. The length of an airbus 747 is about 76 meters. Therefore, 1 kilometer is approximately the length of 13 Airbuses 747.


