What is extract and raffinate in extraction?
What is extract and raffinate in extraction?
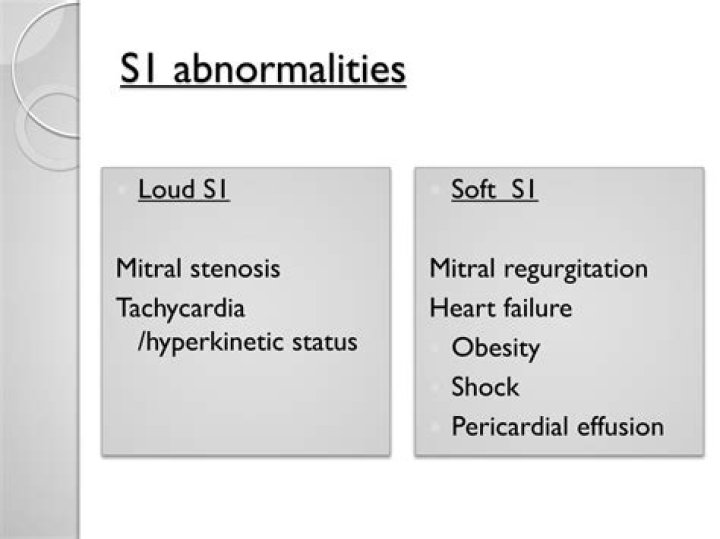
The solvent rich phase is called extract and the residual liquid from which the solute has been removed is called raffinate.
What is the difference between raffinate and extract?
As nouns the difference between raffinate and extract is that raffinate is (chemistry) a solution from which some material has been removed by extraction with an immiscible liquid while extract is that which is extracted or drawn out.
What is extract in solvent extraction?
Solvent extraction is a process in which compounds are separated based on their relative solubilities. This treatment method involves using a solvent – a fluid that has the ability to dissolve another substance. Solvent extraction is used to separate hazardous contaminants from sludge and sediments as well.
What is the equipment used for solvent extraction?
Types include liquid-liquid extractors, continuous extractors, extraction thimbles, extractors with stopcocks, jacketed extractors, fritted extraction thimbles, digestion apparatus & extraction tubes. Extraction equipment is available in different configurations & sizes.
What is raffinate used for?
Raffinate 1 is a chemical building block used in the manufacture of methyl tertiary butyl ether (MTBE) and diisobutylene (DIB). MTBE is a liquid added to petroleum to reduce emissions, and DIB is an intermediate in the production of alcohols and solvents.
What raffinate means?
Definition of raffinate : a liquid product resulting from extraction of a liquid with a solvent also : the less soluble residue that remains after extraction (as in refining lubricating oil)
What is the meaning of raffinate?
How do you extract solvents?
Solvent extraction is the most widely used method. The extraction of natural products progresses through the following stages: (1) the solvent penetrates into the solid matrix; (2) the solute dissolves in the solvents; (3) the solute is diffused out of the solid matrix; (4) the extracted solutes are collected.
What is the difference between extract and tincture?
The difference between extracts and tinctures is the amount of herb infused in the alcohol or glycerin. An extract is considered 1 part herb to 1 part alcohol or glycerin. A tincture is considered 1 part herbs to 3 parts alcohol or glycerin.
What are extraction equipments?
In the chemical industry, three main types of extraction equipment are in use:
- mixer-settler extractors.
- extraction columns and.
- centrifugal extraction equipment.
Which instrument is used for extraction method?
Liquid-liquid extractions in the laboratory usually make use of a separatory funnel, where two immiscible phases are combined to separate a solute from one phase into the other, according to the relative solubility in each of the phases.
How is raffinate made?
Raffinate 1 is a mixture of butenes (iso-butenes and n-butenes) with butane and residual butadiene. Raffinate 2 is produced from Raffinate 1 after iso-butenes have been extracted. It consists of a mixture of n-butenes, butane and potentially some residual butadiene.
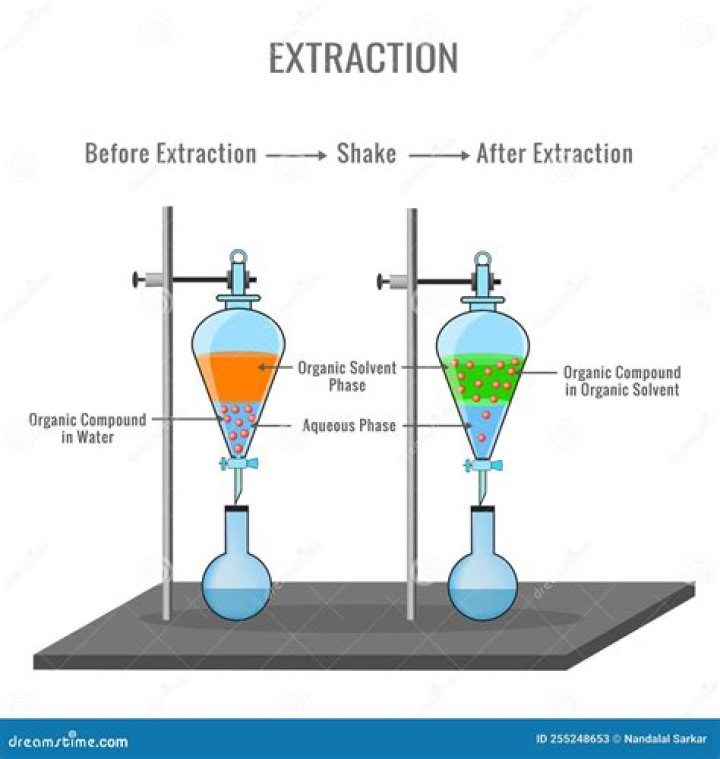
After the extraction of the product the feed liquor is called raffinate (blue liquid with less white dots) whereas the solvent containing the product is called extract (yellow liquid with white dots). For the recovery of the product the solvent has to be separated in a subsequent third step from the product which is mostly done by distillation.
What is the role of solvent in the extraction process?
In a sense, the role of solvent in extraction is analogous to the role of enthalpy in distillation. The solvent-rich phase is called the extract, and the carrier-rich phase is called the raffinate. A high degree of separation may be achieved with several extraction stages in series, particularly in countercurrent flow.
What is a raffinate solution?
A decoction, solution, or infusion made by drawing out from any substance that which gives it its essential and characteristic virtue In chemical separation terminology, the raffinate (from French raffiner, to refine) is a product which has had a component or components removed.
What is the first step in the extraction process?
The first step of an extraction process is mixing for an intensive contact of both liquid phases to enable the mass transfer of the product (white dots) from the (blue) feed liquor into the (yellow) solvent. The second step is the phase separation or settling of the 2 liquid phases.