What is difference between phenyl and benzene?
.
Keeping this in view, what is the difference between phenyl and benzyl?
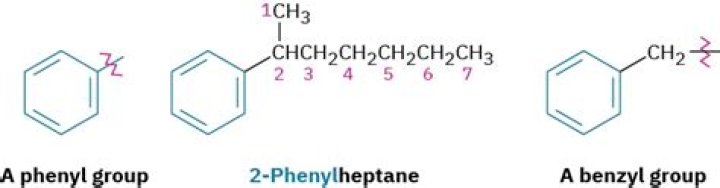
Difference Between Phenyl and Benzyl. Both phenyl and benzyl are derived from benzene, and commonly confused by students of chemistry. Phenyl is a hydrocarbon molecule with the formula C6H5, whereas benzyl is C6H5CH2; an extra CH2 group attached to the benzene ring.
Also, is phenyl an alkyl group? An example of an alkyl phenyl you might see often in class is a benzyl group, like in benzyl bromide (bromomethylbenzene). For this group, n=1 . You can see an indolyl group in tryptophan, the only natural aromatic amino acid with two rings.
Herein, is benzene and benzyl the same?
The terms phenyl and phenol, along with benzene and benzyl, can confuse beginning organic chemistry students. The phenyl group is based simply on benzene, with one H removed. The benzyl group is based on methylbenzene (toluene), with one H removed from the methyl group.
Is benzene a phenyl group?
Phenyl group. In organic chemistry, the phenyl group or phenyl ring is a cyclic group of atoms with the formula C6H5. Phenyl groups are closely related to benzene and can be viewed as a benzene ring, minus a hydrogen, which may be replaced by some other element or compound to serve as a functional group.
Related Question AnswersWhat is phenyl used for?
Phenyl is a strong deodorant and germicide for disinfecting areas covering places like hospital, nursing homes, drains, lavatory, toilets, and cowsheds and is extensively used for sanitation purpose.Why is benzene called phenyl?
As this rule suggests that the benzene ring will act as a function group (a substituent) whenever a substituent of more than six (6) carbons is attached to it, the name "benzene" is changed to phenyl and is used the same way as any other substituents, such as methyl, ethyl, or bromo.Is benzyl a functional group?
Benzyl is a functional group, consisting of a benzene ring, attached to a CH2 group. Its chemical formula is C6H5CH2–. For example, the benzyl alcohol can be marked as BnOH. The position of the first C atom bonded to an aromatic ring is described as benzylic.Is benzene a functional group?
Benzene ring: An aromatic functional group characterized by a ring of six carbon atoms, bonded by alternating single and double bonds. A benzene ring with a single substituent is called a phenyl group (Ph).How do you make phenyl?
Add 1 liter of concentrated white phenyl to the water. Measure out one liter of the concentrated solution and add it to the 39 liters of water. Stir the solution well. The solution should appear milky while stirring.How do you name Esters?
Esters can be named using a few steps Esters are named as if the alkyl chain from the alcohol is a substituent. No number is assigned to this alkyl chain. This is followed by the name of the parent chain from the carboxylic acid part of the ester with an –e remove and replaced with the ending –oate.Is phenyl a functional group?
Phenyl is a functional group with an aromatic ring bonded to another group. And, phenol is a molecule that is just a phenyl bonded to a hydroxyl group. However, some sources consider phenol itself a functional group.Can we die by drinking phenyl?
"His hands and feet had turned gangrenous and it finally resulted in his death,'' a doctor said. "This is called corrosive poisoning and can cause severe damage to internal organs,'' he added.Is phenyl dangerous to health?
* Breathing Phenyl Acetonitrile can irritate the nose, throat and lungs causing coughing, wheezing and/or shortness of breath. * High exposure to Phenyl Acetonitrile can cause headache, nausea, vomiting, weakness, and confusion.Why is benzene carcinogenic?
Because benzene is a volatile aromatic solvent, inhalation exposures predominate. The carcinogenic properties of benzene have long been recognized; an increased risk of leukemia has been shown in workers exposed to high levels of benzene. Benzene exposure is associated with myelodysplastic syndromes.Is phenyl ortho para directing?
Type 3- Alkyl groups are ortho, para-directing groups. whereas -CX3,-(NR3)+ are meta--directing groups. The directive influence can easily be decided by drawing the resonating structures of the intermediate after ortho, meta or para - attack and comparing their stabilities.How do you pronounce phenyl?
Here are 4 tips that should help you perfect your pronunciation of 'phenyl':- Break 'phenyl' down into sounds: [FEE] + [NYL] - say it out loud and exaggerate the sounds until you can consistently produce them.
- Record yourself saying 'phenyl' in full sentences, then watch yourself and listen.


