What is buffer solution pH?
What is buffer solution pH?
A buffer solution (more precisely, pH buffer or hydrogen ion buffer) is an aqueous solution consisting of a mixture of a weak acid and its conjugate base, or vice versa. Buffer solutions are used as a means of keeping pH at a nearly constant value in a wide variety of chemical applications.
How is the pH of a buffer solution determined?
The pH of a buffer is determined by two factors; 1) The equilibrium constant Ka of the weak acid and 2) the ratio of weak base [A-] to weak acid [HA] in solution. If a buffer has more base than acid, more OH- ions are likely to be present and the pH will rise.
What is a buffer solution simple definition?
A buffer is a solution that can resist pH change upon the addition of an acidic or basic components. It is able to neutralize small amounts of added acid or base, thus maintaining the pH of the solution relatively stable.
What is buffer solution give an example?
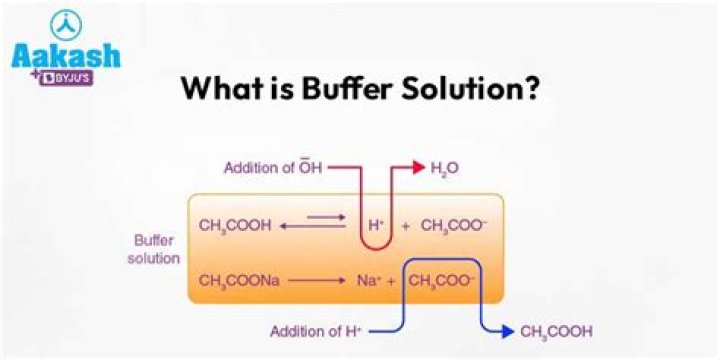
Buffer solutions resist a change in pH when small amounts of a strong acid or a strong base are added. A solution of acetic acid and sodium acetate CH3COOH + CH3COONa is an example of a buffer that consists of a weak acid and its salt.
What is the difference between pH and buffer pH?
pH is a fundamental scale that we use in chemistry to measure the acidity r basicity of a solution. Buffers are chemical solutions that can resist the changes in pH. Therefore, the difference between pH and buffer is that the pH is a logarithmic scale whereas a buffer is an aqueous solution.
What is pH formula?
pH Calculation Formula The formula to calculate pH is: pH = -log[H+] The brackets [] refer to molarity, M. Molarity is given in units of moles per liter of solution. In a chemistry problem, you may be given concentration in other units.
What is the main function of a buffer solution?
A buffer is a solution that can resist pH change upon the addition of an acidic or basic components. It is able to neutralize small amounts of added acid or base, thus maintaining the pH of the solution relatively stable. This is important for processes and/or reactions which require specific and stable pH ranges.
What is buffer solution give two example?
A strong base, such as sodium hydroxide solution (NaOH), is added to raise the pH of alkaline buffers….Examples of Buffers.
| Buffer | pKa | pH range |
|---|---|---|
| citric acid | 3.13., 4.76, 6.40 | 2.1 to 7.4 |
| acetic acid | 4.8 | 3.8 to 5.8 |
| KH2PO4 | 7.2 | 6.2 to 8.2 |
| borate | 9.24 | 8.25 to 10.25 |
What is buffer solution and examples?
For example, a buffer can be composed of dissolved acetic acid (HC 2H 3O 2, a weak acid) and sodium acetate (NaC 2H 3O 2, a salt derived from that acid). Another example of a buffer is a solution containing ammonia (NH 3, a weak base) and ammonium chloride (NH 4Cl, a salt derived from that base).
What is buffer solution equation?
Buffer capacity is the capacity of a buffer solution to resist change in its pH. The equation is given by, pH = pKa + log [Salt] / [Acid] The pH of any acidic buffer solution is always less than 7 and the pH of any basic buffer solution is always greater than 7.