What is an example of oxidizing?
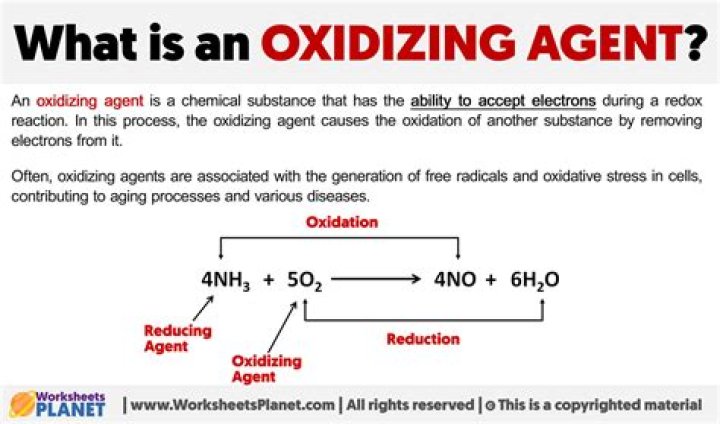
What is an example of oxidizing?
Common examples of oxidizing agents include halogens (such as chlorine and fluorine), oxygen, and hydrogen peroxide (H2O2).
What are three examples of oxidation?
Oxidation Examples The iron metal is oxidized to form the iron oxide known as rust. Meanwhile, oxygen is reduced. Another example of oxidation where an element combines with oxygen is the reaction between magnesium metal and oxygen to form magnesium oxide. Many metals oxidize.
What products are oxidising?
What are some examples of these classes of oxidizing materials?
- aluminum nitrate.
- ammonium persulfate.
- barium peroxide.
- hydrogen peroxide solutions (8% to 27.5% by weight)
- magnesium nitrate.
- nitric acid (40% concentration or less)
- perchloric acid solutions (less than 50% by weight)
- potassium dichromate.
What are types of oxidation?
In this generalized theory, three examples of ways in which oxidation-state changes can occur are by oxygen-atom (gain, oxidation; loss, reduction), hydrogen-atom (loss, oxidation; gain, reduction), and electron (loss, oxidation; gain, reduction) transfer.
What is oxidation reaction give two example?
The reaction in which oxygen combines with other elements or compounds is known as an oxidation reaction. For example, burning of hydrogen is an oxidation process in which hydrogen combines with oxygen to form water. Oxidation reactions are exothermic.
Is Acid an oxidising agent?
Most Brønsted acids can act as oxidizing agents, because the acidic proton can be reduced to hydrogen gas. Some acids contain other structures that act as stronger oxidizing agents than hydrogen ions. These include nitric acid, perchloric acid, chloric acid, chromic acid, and concentrated sulfuric acid, among others.
Is Naocl an oxidizing agent?
Sodium hypochlorite is a strong oxidizer. Oxidation reactions are corrosive.
What is oxidizing agent give two examples?
Common oxidizing agents are oxygen, hydrogen peroxide and the halogens. In one sense, an oxidizing agent is a chemical species that undergoes a chemical reaction in which it gains one or more electrons. In that sense, it is one component in an oxidation–reduction (redox) reaction.
What are the most common oxidizers?
Common oxidizers include Hydrogen peroxide, Nitric acid, Nitrate and Nitrite compounds, Perchloric acid and Perchlorate compounds, and Hypochlorite compounds, such as household bleach.
Is decomposition an example of oxidation?
Decomposition. Similar to the previous example, H2O has a total oxidation state of 0, with each H taking on a +1 state and the O a -2; thus, decomposition oxidizes oxygen from -2 to 0 and reduces hydrogen from +1 to 0.
Why is oxygen an oxidizing agent?
Reduction is gain of electrons. We can split this reaction into two half ionic equations. Hydrogen is losing 4 electrons which Oxygen is gaining. Since oxidation is loss of electrons, H2 is oxidizing to H+ ions and as reduction is gain of electrons O2 is reducing to O- – ions.


