What is an example of an adiabatic process?
What is an example of an adiabatic process?
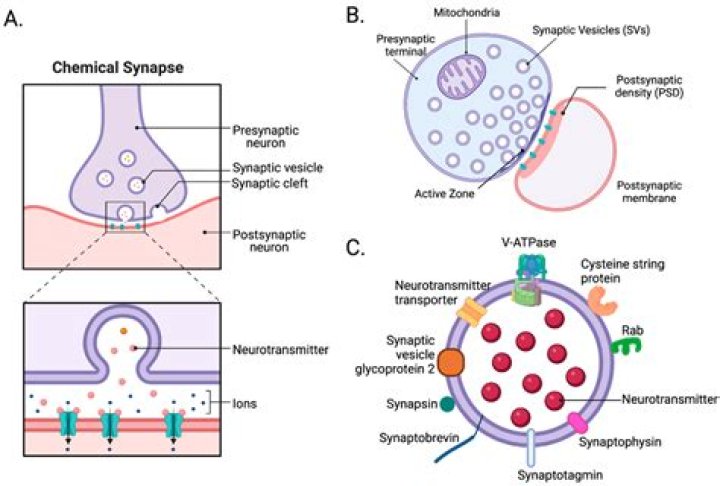
An example of an adiabatic process is the vertical flow of air in the atmosphere; air expands and cools as it rises, and contracts and grows warmer as it descends. Another example is when an interstellar gas cloud expands or contracts. Adiabatic changes are usually accompanied by changes in temperature.
What is adiabatic process in physics?
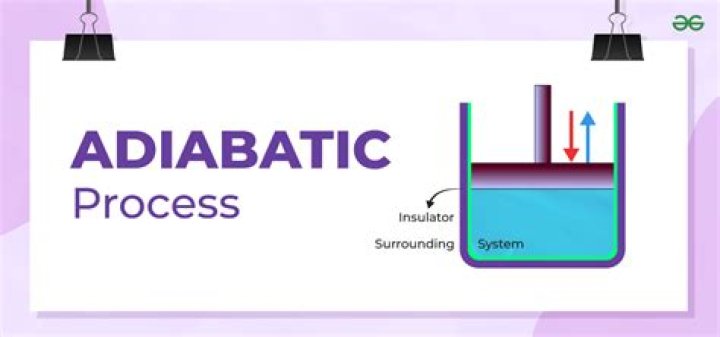
In thermodynamics, an adiabatic process (Greek: adiábatos, “impassable”) is a type of thermodynamic process that occurs without transferring heat or mass between the thermodynamic system and its environment. Unlike an isothermal process, an adiabatic process transfers energy to the surroundings only as work.
How do you calculate work in adiabatic process?
With the adiabatic condition of Equation 3.7. 1, we may write p as K/Vγ, where K=p1Vγ1=p2Vγ2. The work is therefore W=∫V2V1KVγdV=K1−γ(1Vγ−12−1Vγ−11)=11−γ(p2Vγ2Vγ−12−p1Vγ1Vγ−11)=11−γ(p2V2−p1V1)=11−1.40[(1.23×106N/m2)(40×10−6m3)−(1.00×105N/m2)(240×10−6m3)]=−63J.
What happens when a saturated liquid is expanded adiabatically?
THERMODYNAMICS | Saturated Adiabatic Processes The rate at which saturated air cools as it expands adiabatically is smaller than the rate at which unsaturated air cools adiabatically, because part of the cooling is canceled by the latent heat released during condensation.
What are adiabatic operations?
Adiabatic operations are those operations in which neither heat enters or leaves a system. Example:- A system having a perfectly non-conducting boundary.
What is adiabatic expansion of gas?
An adiabatic expansion has less work done and no heat flow, thereby a lower internal energy comparing to an isothermal expansion which has both heat flow and work done. Temperature decreases during adiabatic expansion. The temperature of n moles of an ideal gas changes from to in a quasi-static adiabatic transition.
Are compressors adiabatic?
Reciprocating compressors use pistons to push gas to a higher pressure. When the pressure of a gas is increased in an adiabatic system, the temperature of the fluid must rise. Since the temperature change is accompanied by a change in the specific volume, the work necessary to compress a unit of fluid also changes.
Why does adiabatic expansion causes cooling?
As in this process Workdone is positive(volume increases) so Internal Energy is negative(it decreases) and hence Temperature also decreases. That is why Adiabatic Expansion shows cooling.
Does a gas do any work when it expands adiabatically?
Will the gas do any work? If yes, what will be the source of energy? Does the gas perform any work in adiabatic expansion? What is the source of that energy?
Does temperature increase in adiabatic expansion?
What happens to the temperature of an ideal gas in an adiabatic expansion? An adiabatic expansion has less work done and no heat flow, thereby a lower internal energy comparing to an isothermal expansion which has both heat flow and work done. Temperature decreases during adiabatic expansion.
When gas expands adiabatically the internal energy?
When a real gas expands adiabatically , the decrease in internal energy is equalto the adiabatic work done by the system.
What happens when a saturated vapor is compressed adiabatically?
For compression, adiabatic: If we start with pure saturated steam, without admixture of water, it will be superheated by compression. If the initial steam weight is greater than that of the water, steam is generated by the compression. If there is more water than steam, steam is condensed during compression.