What is an example of a hydrogenation reaction?
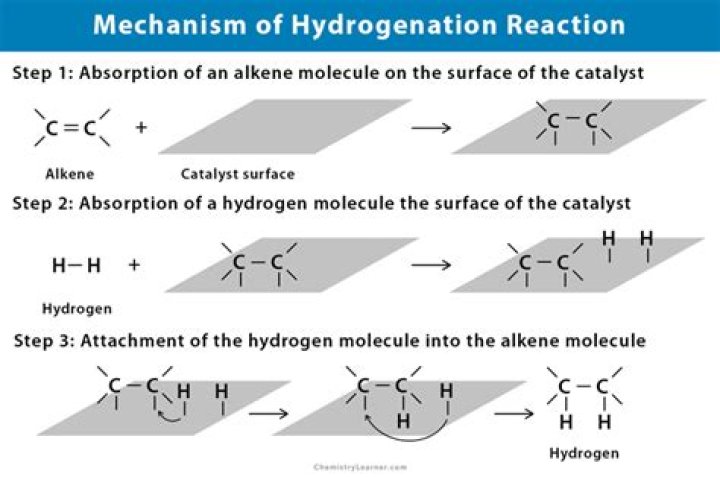
What is an example of a hydrogenation reaction?
Hydrogenation is a chemical reaction that adds hydrogen to a molecule. Hydrogenation is not thermodynamically favorable at ordinary temperatures, so a catalyst is needed. Usually this catalyst is a metal. Examples of hydrogenated products include margarine, mineral turpentine, and aniline.
What is a catalyst in hydrogenation?
Catalytic hydrogenation is treatment with hydrogen in the presence of a catalyst such as nickel, palladium or platinum. Catalysts are required for the reaction to be usable; non-catalytic hydrogenation takes place only at very high temperatures. Hydrogenation reduces double and triple bonds in hydrocarbons.
Which is the most widely used catalyst for hydrogenation?
nickel
3.1 Hydrogenation It is normally accomplished by the high-pressure catalytic reduction of unsaturated double bonds with gaseous hydrogen. The predominant catalyst used commercially is nickel, although recently other metallic and organic catalysts have been used to reduce trans isomerization reactions for food products.
What is hydrogenolysis example?
In petroleum refineries, catalytic hydrogenolysis of feedstocks is conducted on a large scale to remove sulfur from feedstocks, releasing gaseous hydrogen sulfide (H2S). The hydrogen sulfide is subsequently recovered in an amine treater and finally converted to elemental sulfur in a Claus process unit.
Is catalytic hydrogenation syn or anti?
Alkene hydrogenation is the syn-addition of hydrogen to an alkene, saturating the bond. The alkene reacts with hydrogen gas in the presence of a metal catalyst which allows the reaction to occur quickly.
Is catalytic hydrogenation exothermic?
Although the overall hydrogenation reaction is exothermic, a high activation energy prevents it from taking place under normal conditions. This restriction may be circumvented by the use of a catalyst, as shown in the following diagram.
What is catalytic effect?
If you describe a person or thing as having a catalytic effect, you mean that they cause things to happen or they increase the speed at which things happen.
Which catalyst is used in hydrogenation of vegetable oil?
Nickel catalyst
Nickel catalyst is used in commercial hydrogenation of edible oils.
Which catalyst is used in hydrogenolysis?
Hydrogenation is a chemical reaction between molecular hydrogen (H2) and another compound or element, usually in the presence of a catalyst such as nickel, palladium or platinum. The process is commonly employed to reduce or saturate organic compounds.
Does catalytic hydrogenation reduce ketones?
The simplest large-scale procedure for reduction of aldehydes and ketones to alcohols is by catalytic hydrogenation: Hydrogenation of aldehyde and ketone carbonyl groups is much slower than of carbon-carbon double bonds so more strenuous conditions are required. …