What is a non-volatile solution?
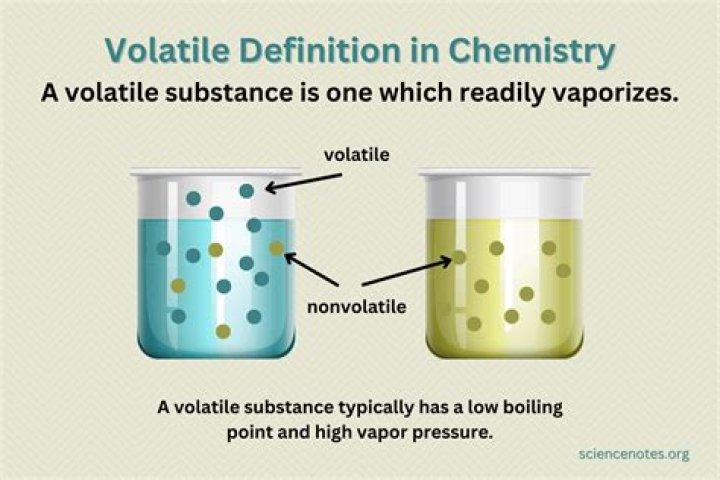
What is a non-volatile solution?
Nonvolatile means that the solute itself has little tendency to evaporate. The solution on the right left has had some of its solvent particles replaced by solute particles. Since the solute particles do not evaporate, the vapor pressure of the solution is lower than that of the pure solvent.
What is non-volatile chemistry?
Nonvolatile is the inability of a substance to readily evaporate into gas. Nonvolatile substances are usually solids with boiling points above 212°F (100°C), and do not vaporize easily at room temperature and pressure.
What is volatile and non-volatile liquid?
A volatile substance is one that evaporates or sublimates at room temperature or below. A non-volatile substance refers to a substance that does not readily evaporate into a gas under existing conditions. Non-volatile substances exhibit a low vapor pressure and a high boiling point.
Is NaCl a non-volatile?
Ethyl alcohol and pentane are examples of volatile substances; sugar and sodium chloride are considered nonvolatile. The presence of a solute leads to a colligative property called the “lowering of the vapor pressure of the solution” when compared to the vapor pressure of the pure solvent.
What is the effect of non volatile impurities?
Nonvolatile impurities can increase the boiling point of the solution. If you add extra substances to the cup of water and sugar, it will take more effort to reach the boiling point. This happens because there are fewer free water molecules that can evaporate, and the partial vapor pressure of the water decreases.
Is Water Non Volatile?
Boiling water. Water (H2O) is moderately volatile. It has a boiling point of 100oC and evaporates only slowly at room temperature. It is not flammable or explosive.
What is the difference between a volatile and non volatile solute?
A non-volatile solute does not produce vapour at the boiling point of the solution. A volatile solute does produce vapour at the boiling point of the solution.
What is non volatile material?
Learn about our Editorial Process. Updated on October 06, 2019. In chemistry, the term nonvolatile refers to a substance that does not readily evaporate into gas under existing conditions. In other words, a nonvolatile material exerts a low vapor pressure and has a slow rate of evaporation.
Is urea Non-volatile?
The weight in g, of the non-volatile solute urea(NH2CONH2) to be dissolved in 100 g of water in order to decreases its vapour pressure by 1.
Is sucrose non-volatile?
Sugar (sucrose) and salt (sodium chloride) are nonvolatile solids. It’s probably easier to imagine a nonvolatile substance if you consider the properties of materials that are volatile. Examples include alcohol, mercury, gasoline, and perfume. Volatile substances readily release their molecules into the air.
What is the effect of the presence of a non volatile impurity on the observed boiling point?
What is Raoult’s Law for non volatile solute?
Raoult’s law – The vapour pressure of a solution of a non-volatile solute is equal to the vapour pressure of the pure solvent at that temperature multiplied by its mole fraction.