What is a cooling curve experiment?
What is a cooling curve experiment?
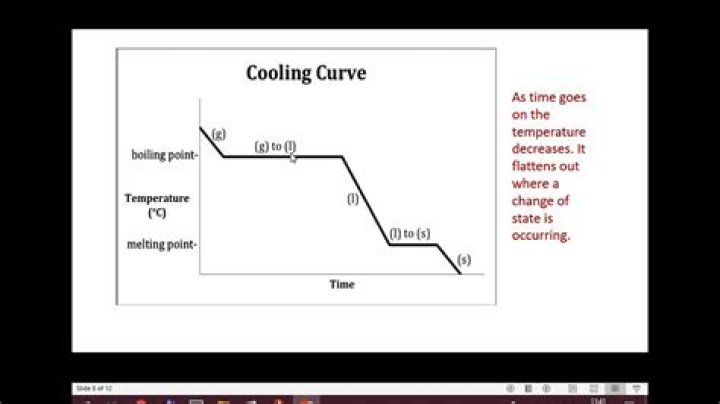
A cooling curve of a substance is a graph of the variation of the temperature with time as it is allowed to cool. The gradient of the cooling curve is related to the heat capacity, the thermal conductivity of the substance, and the external temperature.
What are cooling curves used for?
A cooling curve is a type of graph used in chemistry, physics, engineering, and other disciplines to chart the progress of a cooling substance. One axis of graph, usually the x axis, charts time, while temperature is represented on the other axis.
How are cooling curves used in industry?
The cooling curve serves as a “finger print” of the solidification process and can be used to predict the structure of the test sample and consequently the actual casting.
Why are cooling curves used?
A cooling curve is a line graph that represents the change of phase of matter, typically from a gas to a solid or a liquid to a solid. This is because the matter has more internal energy as a liquid or gas than in the state that it is cooling to. The amount of energy required for a phase change is known as latent heat.
What is the difference between a heating and cooling curve?
Heating curves show how the temperature changes as a substance is heated up. Cooling curves are the opposite. They show how the temperature changes as a substance is cooled down.
What is supercooling in cooling curve?
Supercooling, also known as undercooling, is the process of lowering the temperature of a liquid or a gas below its freezing point without it becoming a solid. It achieves this in the absence of a seed crystal or nucleus around which a crystal structure can form.
What are cooling curves in engineering materials and heat treatment?
to extract heat. Cooling curves provide a complete picture of the heat extraction and cooling performance of a quenchant, as a function of surface temperature or center temperature of a probe.
How does cooling occur in water?
When a hotter particle near the surface of the liquid ‘breaks free’ from it’s liquid bonds, it escapes as a gas, carrying away energy from the rest of the liquid. The average energy of the liquid therefore decreases – in other words, the liquid cools down.
Why is glass called a supercooled liquid?
Glass is called supercooled liquid because glass is an amorphous solid. Amorphous solids have the tendency to flow but, slowly. It does not form a crystalline solid structure as particles in solids do not move but here it moves. Hence it is called a supercooled liquid.