What is a common cause of incomplete combustion?
.
Similarly, you may ask, which is a common cause of incomplete combustion Brainly?
Combustion is a chemical reaction in which a fuel is reacted with excess supply of oxygen to result into complete oxidation of fuel to give carbon dioxide and water. Incomplete combustion happens when the supply of oxygen is limited and thus fuel can not be completely oxidized , thus forming carbon monoxide.
Likewise, why is incomplete combustion harmful to humans? The incomplete combustion of hydrocarbons produces carbon monoxide, which is a poisonous and potentially fatal gas to humans. Carbon monoxide reduces hemoglobin's (a pigment/protein in our blood that carries oxygen) ability to carry oxygen around our body, essentially starving our organs of oxygen.
Beside this, what are the products of incomplete combustion?
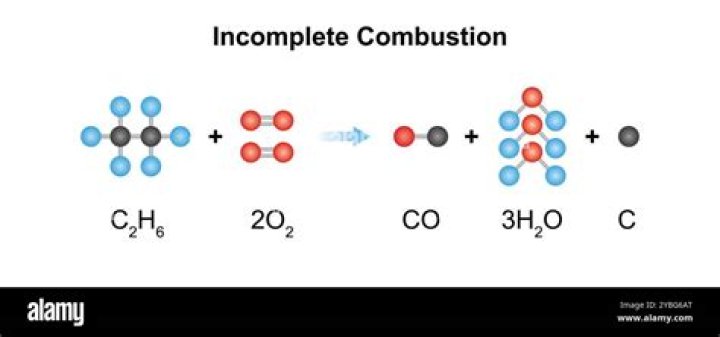
Incomplete combustion is also a reaction between oxygen and fuel but the products are carbon monoxide, water and carbon. Incomplete combustion occurs when a combustion reaction occurs without a sufficient supply of oxygen.
Which gas is evolved by incomplete combustion of wood or coal?
Incomplete combustion of fuel takes place when there is not enough oxygen supplied to it. Due to lack of oxygen, Carbon monoxide is formed as a result of incomplete combustion which is a poisonous gas. When complete combustion takes place, carbon dioxide is produced.
Related Question AnswersWhich chemical equation shows the incomplete combustion of methane?
Which chemical equation shows the incomplete combustion of methane? A. C3H8 + 5O2 → 3CO2 + 4H2O. B.What does insufficient combustion of carbon produces mention its impact?
Answer: During incomplete combustion of hydrocarbons/carbon carbon monoxide, water and carbon are produced as byproducts instead of CO2. Insufficient combustion usually takes place when the supply of air or oxygen is inadequate. In this reaction, carbon is produced as soot.What might be the environmental consequences of insufficient oxygen were present for the combustion reaction?
Problems associated with incomplete combustion Environmental problems: These oxides combine with water and oxygen in the atmosphere, creating nitric acid and sulfuric acids, which return to Earth's surface as acid deposition, or "acid rain." Acid deposition harms aquatic organisms and kills trees.What is an example of incomplete combustion?
An example of incomplete combustion would be burning coal (a fossil fuel), during which quantities of soot and carbon monoxide are released. In fact, many fossil fuels—including coal—burn incompletely, releasing waste products into the environment.What are the effects of incomplete combustion?
There are various impacts of combustion on the environment, these impacts can be caused by; Gas leaks, oil spillage, noise and air pollution. Incomplete combustion of hydrocarbons also results in carbon monoxide pollution. An odorless, colorless gas, carbon monoxide can be harmful to both the environment and to people.How do you identify incomplete combustion?
Combustion requires three things to occur: fuel (hydrocarbons), oxygen (from the air), and a catalytic spark. Complete combustion will produce only carbon dioxide and water as the products and nothing will be leftover. Incomplete combustion will produce other byproducts like carbon monoxide or carbon soot left behind.What is difference between complete and incomplete combustion?
Complete combustion occurs when there is enough oxygen to completely use up all the reactants. Complete combustion produces carbon dioxide that adds to global warming while incomplete combustion produces carbon monoxide that is toxic. Incomplete combustion also produces smoke which contributes to air pollution.What is complete and incomplete combustion?
Hydrocarbon fuels can undergo complete combustion or incomplete combustion, depending on the amount of oxygen available. Complete combustion happens when there is a good supply of air. Carbon and hydrogen atoms in the hydrocarbon fuel react with oxygen in an exothermic reaction: carbon dioxide and water are produced.What gases are contained in the products of incomplete combustion?
Fossil fuels contain carbon (C) and hydrogen (H). During complete combustion carbon and hydrogen combine with oxygen (O2) to produce carbon dioxide (CO2) and water (H2O). During incomplete combustion part of the carbon is not completely oxidized producing soot or carbon monoxide (CO).What are the products of perfect combustion?
We will now look at six products of combustion:- Carbon Dioxide.
- Carbon Monoxide.
- Sulfur Dioxide.
- Nitrogen Oxides.
- Lead.
- Particulate Matter.
How do you test for products of complete combustion?
Products of combustion- When a compound burns it reacts with oxygen.
- The products from the combustion of hydrocarbon fuels can be identified with the following set up in the lab.
- Carbon dioxide gas turns limewater from colourless to milky white.
- If water is produced it will condense in the u tube cooled by the ice water.


