What happens when magnesium hydroxide reacts with hydrochloric acid?
What happens when magnesium hydroxide reacts with hydrochloric acid?
a) The reaction between magnesium hydroxide and hydrochloric acid is an example of neutralisation reaction. The hydroxide and hydrogen ions will combine to form water. The remaining ions combine to produce the salt of neutralisation. The salt is magnesium chloride.
When hydrochloric acid reacts with magnesium hydroxide it gives magnesium chloride and water?
Mg(OH)2+HCl–>MgCl2+H2O.
What type of reaction is Mg OH 2 HCl → MgCl2 H2O?
Balancing Strategies: This is a neutralization reaction, and can also called a double displacement reaction. It’s neutralization because we have a base, the magnesium hydroxide, added to an acid, HCl. The products are a salt (MgCl2) and and water.
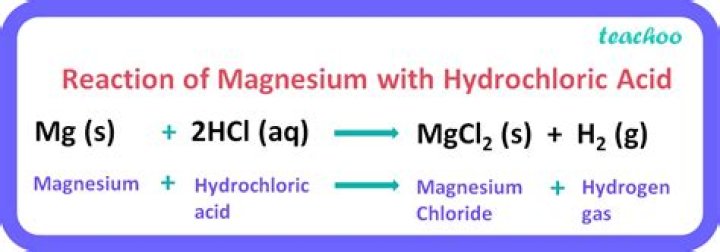
What type of reaction is magnesium and hydrochloric acid?
single replacement reaction
The reaction between magnesium and hydrochloric acid combine to form a salt of magnesium chloride and release hydrogen gas. This single replacement reaction is a classic example of a metal reacting in an acid to release hydrogen gas. I hope this was helpful.
What is the word equation for magnesium and hydrochloric acid?
Magnesium reacts with hydrochloric acid according to the equation: Mg(s) + 2 HCl(aq) –> MgCl 2(aq) + H 2(g) This demonstration can be used to illustrate the characteristic reaction of metals with acid, a single replacement reaction, or to demonstrate the generation of hydrogen gas.
What products are formed when hydrochloric acid HCL and magnesium hydroxide Mg OH 2 react in an acid base reaction?
Neutralization reaction between magnesium hydroxide and hydrochloric acid Mg(OH)2(s) + 2HCl(aq) → 2H2O(l) + MgCl2(aq) b.
What type of reaction is magnesium and HCL?
single replacement
The reaction between magnesium and hydrochloric acid combine to form a salt of magnesium chloride and release hydrogen gas. This reaction is an example of a single replacement. This single replacement reaction is a classic example of a metal reacting in acid to release hydrogen gas.
What is MgCl2 H2O?
Cl2H2MgO. Synonyms. Magnesium chloride, monohydrate. magnesium dichloride monohydrate. MgCl2.H2O.
What happens when magnesium reacts with water?
Magnesium. Magnesium burns in steam to produce white magnesium oxide and hydrogen gas. Very clean magnesium ribbon has a mild reaction with cold water, given below. After several minutes, hydrogen gas bubbles form on its surface, and the coil of magnesium ribbon usually floats to the surface.
What type of reaction is Pb NO3 2 and Ki?
metathesis reaction
The reaction is an example of a metathesis reaction, which involves the exchange of ions between the Pb(NO3)2 and KI.


