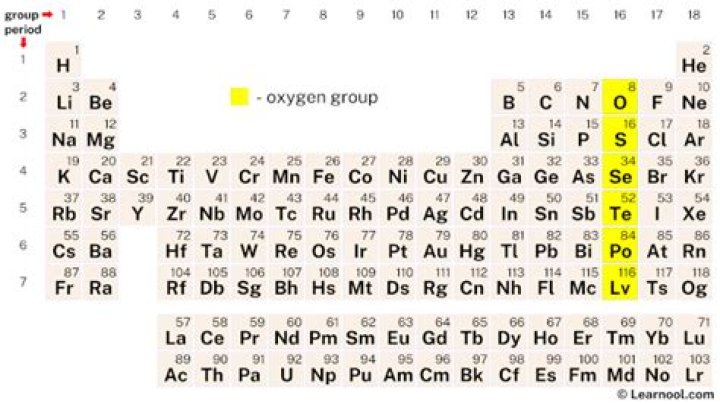
What group and period is oxygen in
Group16−218.79°C, −361.82°F, 54.36 KPeriod2−182.962°C, −297.332°F, 90.188 KBlockp0.001308Atomic number815.999State at 20°CGas16O
What is Group 17 called in the periodic table?
halogen, any of the six nonmetallic elements that constitute Group 17 (Group VIIa) of the periodic table. The halogen elements are fluorine (F), chlorine (Cl), bromine (Br), iodine (I), astatine (At), and tennessine (Ts).
What is Group 18 called?
noble gas, any of the seven chemical elements that make up Group 18 (VIIIa) of the periodic table. The elements are helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), radon (Rn), and oganesson (Og).
What are the elements in group 15?
nitrogen group element, any of the chemical elements that constitute Group 15 (Va) of the periodic table. The group consists of nitrogen (N), phosphorus (P), arsenic (As), antimony (Sb), bismuth (Bi), and moscovium (Mc).Which is the 18th group of periodic table?
Atomic NumberSymbolName18ArArgon19KPotassium20CaCalcium21ScScandium
What element is in group 18 Period 1?
Group118Atomic # Name1 H2 He
What is the group name for oxygen?
oxygen group element, also called chalcogen, any of the six chemical elements making up Group 16 (VIa) of the periodic classification—namely, oxygen (O), sulfur (S), selenium (Se), tellurium (Te), polonium (Po), and livermorium (Lv).
What is a group of 14 called?
Why is group 14 called crystallogens? – Quora.What is a main group element in Period 5?
The period 5 transition metals are yttrium (Y), zirconium (Zr), niobium (Nb), molybdenum (Mo), technetium (Tc), ruthenium (Ru), rhodium (Rh), palladium (Pd), silver (Ag), and cadmium (Cd).
What is the name of AU?Atomic NumberSymbolName79AuGold72HfHafnium108HsHassium2HeHelium
Article first time published onWhy is Group 17 called halogens?
The group 17 elements include fluorine(F), chlorine(Cl), bromine(Br), iodine(I) and astatine(At) from the top to the bottom. They are called “halogens” because they give salts when they react with metals.
What are group and period in the periodic table?
The columns of the periodic table are called groups. Members of the same group in the table have the same number of electrons in the outermost shells of their atoms and form bonds of the same type. The horizontal rows are called periods. … He proposed the periodic law and devised the tabular arrangement of elements.
Which element is in period 3 Group 17?
Chlorine is a halogen in group 17 and period 3. It is very reactive and is widely used for many purposes, such as as a disinfectant. Due to its high reactivity, it is commonly found in nature bonded to many different elements.
What is the family or group of oxygen?
Group 16 is the oxygen family. It consists of the elements oxygen, sulfur, selenium, tellurium, and polonium. Each has six of the desired eight electrons required for the octet in its highest energy level.
Why is oxygen and sulfur in the same group?
Sulfur forms many compounds that are analogues of Oxygen compounds. So, Oxygen and Sulphur are kept in the same group, the reason is a similar number of electrons in the outermost shell.
Is oxygen a compound?
Oxygen is a chemical element with symbol O and atomic number 8. Classified as a nonmetal, Oxygen is a gas at room temperature.
What element is in Group 13 and Period 2?
The boron family contains elements in group 13 of the periodic talbe and include the semi-metal boron (B) and the metals aluminum (Al), gallium (Ga), indium (In), and thallium (Tl).
What element is in period 3 Group 13?
Element#SymbolAluminium13AlSilicon14SiPhosphorus15PSulfur16S
Which element is in Group 3 Period 5?
PeriodGroup2Li 3C 63Na 11Si 144K 19Ge 325Rb 37Sn 50
Is oxygen a main group element?
The oxygen family, also called the chalcogens, consists of the elements found in Group 16 of the periodic table and is considered among the main group elements. It consists of the elements oxygen, sulfur, selenium, tellurium and polonium. These can be found in nature in both free and combined states.
What is the group number of the element oxygen?
Group16−218.79°C, −361.82°F, 54.36 KPeriod2−182.962°C, −297.332°F, 90.188 KBlockp0.001308Atomic number815.999State at 20°CGas16O
What element is in group 14 Period 5?
Tin. Tin is a chemical element with the symbol Sn (for Latin: stannum) and atomic number 50. It is a main-group metal in group 14 of the periodic table.
What is the name for a group of 10?
As a matter of fact, ‘decade‘ does mean ‘set of ten’ and is used also in contexts other than years.
What are groups of 12 called?
Dozen, a grouping of twelve.
What is Group 13 called?
IUPAC group number13Name by elementboron groupTrivial nametrielsCAS group number (US, pattern A-B-A)IIIAold IUPAC number (Europe, pattern A-B)IIIB
Why is gold named gold?
Discovery dateapprox 3000BCDiscovered by-Origin of the nameThe name is the Anglo-Saxon word for the metal and the symbol comes from the Latin ‘aurum’, gold.
Is gold a real color?
Gold, also called golden, is a color. The web color gold is sometimes referred to as golden to distinguish it from the color metallic gold. … Metallic gold, such as in paint, is often called goldtone or gold tone, or gold ground when describing a solid gold background.
How did gold get on earth?
All of the gold found on Earth came from the debris of dead stars. As the Earth formed, heavy elements such as iron and gold sank toward the planet’s core. If no other event had occurred, there would be no gold in the Earth’s crust. But, around 4 billion years ago, Earth was bombarded by asteroid impacts.
Why are Group 16 elements called Chalcogens?
-Group-16 elements are also called chalcogens. They are called so because most of the copper ores have copper in the form of oxides and sulphides. They also contain small amounts of selenium and tellurium. The ores of copper are called ‘chalcos’ in Greek.
Why are Group 16 called halogens?
They are called “halogens” because they give salts when they react with metals. The valence shell electronic configuration of these electrons is ns2np5. … The electron gain enthalpy of these elements becomes less negative upon moving down the group. Fluorine has lesser enthalpy than chlorine.
What is the group name of boron?
boron group element, any of the six chemical elements constituting Group 13 (IIIa) of the periodic table. The elements are boron (B), aluminum (Al), gallium (Ga), indium (In), thallium (Tl), and nihonium (Nh).


