What are van der Waals heterostructures?
What are van der Waals heterostructures?
While strong covalent bonds provide in-plane stability of the 2D crystals, these materials are called van der Waals heterostructures because the atomically thin layers are not mixed through a chemical reaction but rather attached to each other via a weak so called van der Waals interaction – similar to how a sticky …
Is graphene a van der waals?
Bonding forces of graphene are very weak, comparable with these in molecu- lar crystals. Such forces in molecular crystals are the van der Waals forces resulting from the induced polarity. Because of weak forces between graphenes it was as- sumed that they are the van der Waals forces.
How is boron nitride made?
Boron nitride is produced synthetically. Hexagonal boron nitride is obtained by the reacting boron trioxide (B2O3) or boric acid (H3BO3) with ammonia (NH3) or urea (CO(NH2)2) in a nitrogen atmosphere: B2O3 + 2 NH3 → 2 BN + 3 H2O (T = 900 °C) B(OH)3 + NH3 → BN + 3 H2O (T = 900 °C)
What is a van der Waals material?
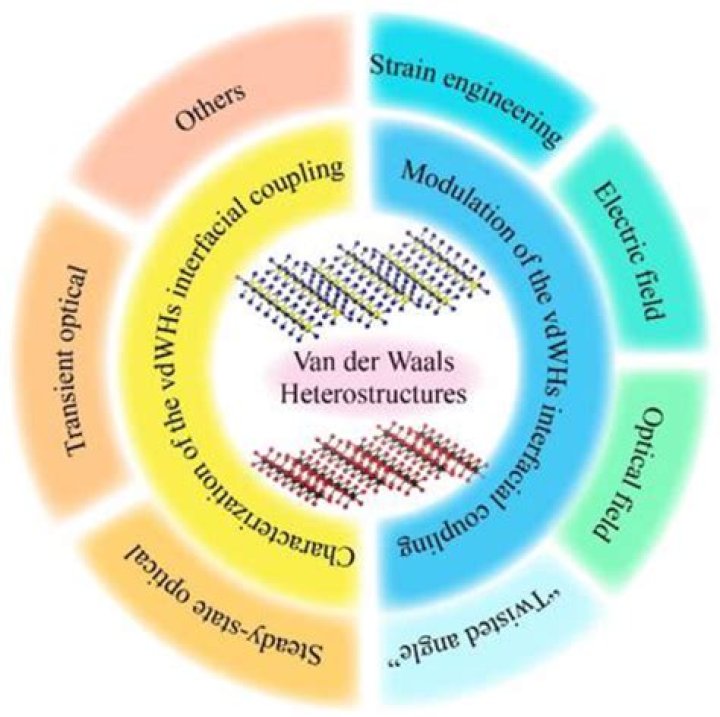
Van der Waals materials, i.e. materials with strong in-plane covalent bonding and weak interlayer (van der Waals) interactions, have become a focal area for materials research. Given the two-dimensional nature of these materials, interfaces naturally play a crucial role in controlling or modifying their properties.
What is van der Waals crystal?
The van der Waals force binds atoms and molecules within a few angstroms of each other; beyond that distance the force is negligible. The molecules attract one another by van der Waals forces, which provide the crystal binding.
What are van der Waals materials?
Van der Waals (vdW) materials are made up of strongly bonded two-dimensional (2D) layers that are bound in the third dimension through weaker dispersion forces. Graphite is a vdW material broadly used in industry in electrodes, lubricants, fibers, heat exchangers, and batteries.
How expensive is boron nitride?
Hexagonal Boron Nitride (HBN) Powder Order Page – All Sizes
| Quantity | Price |
|---|---|
| 3-9 Items | $64.00 |
| 10+ Items | $54.00 |
What is the hardest substance on earth?
diamond
In diamond, these electrons are shared with four other carbon atoms to form very strong chemical bonds resulting in an extremely rigid tetrahedral crystal. It is this simple, tightly-bonded arrangement that makes diamond one of the hardest substances on Earth.
What are Vander Waals solids?
A molecular solid is a solid consisting of discrete molecules. van der Waals) and electronic characteristics (i.e. delocalized electrons) from other types of solids give rise to the unique mechanical, electronic, and thermal properties of molecular solids.
What is an example of a van der Waals force?
Examples of van der Waals forces include hydrogen bonding, dispersion forces, and dipole-dipole interactions.
What molecules are van der Waals?
WHAT ARE VAN DER WAALS MOLECULES? Van der Waals molecules are weakly bound complexes of small atoms or molecules held together, not by chemical bonds, but by intermolecular attractions. Gaseous argon, for example, at lOooK, contains in addition to Ar atoms a small concentration of the Ar 2 van der Waals molecule.