What are the units of pressure in chemistry?
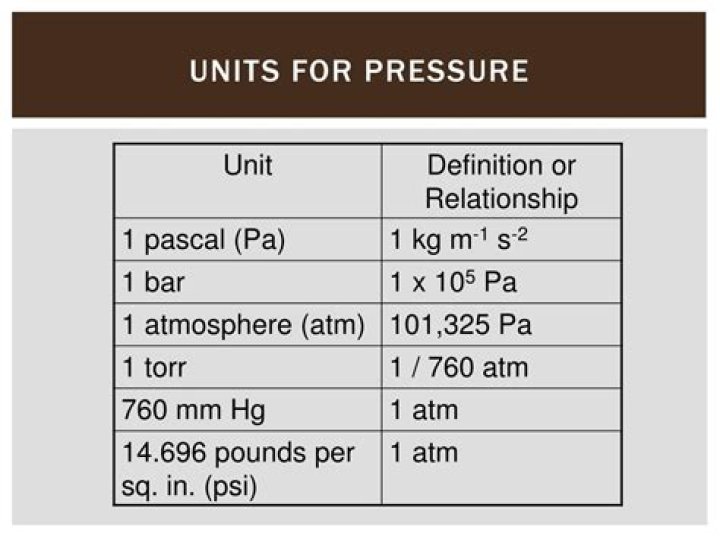
What are the units of pressure in chemistry?
The unit of pressure in the SI system is the pascal (Pa), defined as a force of one Newton per square meter. The conversion between atm, Pa, and torr is as follows: 1 atm = 101325 Pa = 760 torr. A standardized prefix system indicates fractions and multiples of metric units (e.g. milli-, mega-).
What are the 4 main units of pressure in chemistry?
Pressure is defined as a measure of the force applied over a unit area. Pressure is often expressed in units of Pascals (Pa), newtons per square meter (N/m2 or kg/m·s2), or pounds per square inch. Other units include the atmosphere (atm), torr, bar, and meters sea water (msw).
What is pressure and its units?
Pressure is defined as the physical force exerted on an object. The basic formula for pressure is F/A (Force per unit area). Unit of pressure is Pascals (Pa). Types of Pressures are Absolute, Atmospheric, Differential, and Gauge Pressure.
What are the 5 unit of pressure?
5 units of pressure are bar, atm, Pascal, kilo Pascal and torr. Pressure is defined as the perpendicular force which is applied in an unit area.
What is the derived unit of pressure?
pascal
A pascal is the SI-derived unit of measurement for pressure. The pascal is one newton (an SI-derived unit itself) per square meter.
What are the 7 units of pressure?
The SI unit for pressure is pascals (Pa). Other units of pressure include torr, barr, atm, at, ba, psi, and manometric units like mm Hg and fsw.
What is meant by 1 torr of pressure?
The torr (symbol: Torr) is a unit of pressure based on an absolute scale, defined as exactly 1760 of a standard atmosphere (101325 Pa). Thus one torr is exactly 101325760 pascals (≈ 133.32 Pa).
What are the 5 units of pressure?
How do we measure pressure?
Pressure is typically measured in units of force per unit of surface area ( P = F / A). In physical science the symbol for pressure is p and the SI unit for measuring pressure is pascal (symbol: Pa). One pascal is the force of one Newton per square meter acting perpendicular on a surface.
Which is the biggest pressure unit?
Rank the common units of pressure in order of smallest to largest unit: 1 atm, 1 psi, 1 torr, 1 mm Hg, 1 Pa, 1 in Hg.
What is pressure write the SI unit of pressure?
Pressure is defined as force applied per unit area. Its unit in SI system is Newton per meter square (or) Pascal.
Why is mmHg the same as torr?
Pressure Units and Conversion A barometer measures gas pressure by the height of the column of mercury. One unit of gas pressure is the millimeter of mercury (mmHg). An equivalent unit to the mmHg is called the torr, in honor of the inventor of the barometer, Evangelista Torricelli.