What are the units of a reaction rate?
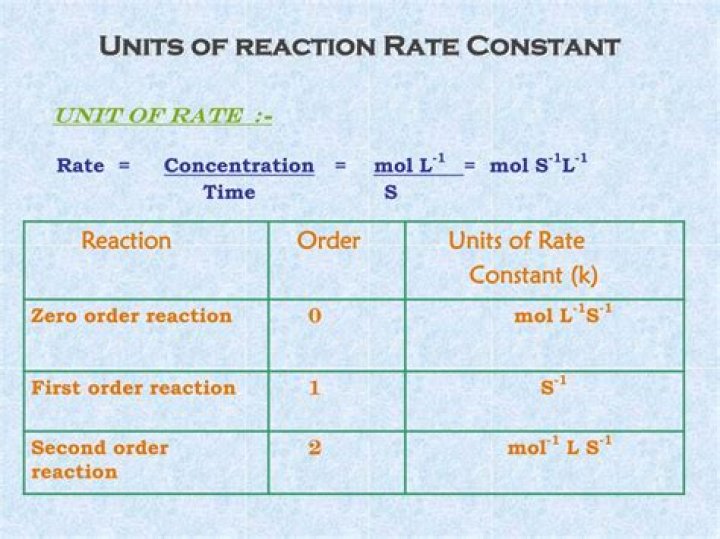
What are the units of a reaction rate?
Reaction rates are usually expressed as the concentration of reactant consumed or the concentration of product formed per unit time. The units are thus moles per liter per unit time, written as M/s, M/min, or M/h.
How does the amount of catalyst affect reaction rate?
Use of catalyst increases rate of reaction. A catalyst lowers the activation energy and so more particles can attain activation energy, thus a faster rate of reaction. Only a small amount of catalyst is required. Increasing the amount of catalyst used will not increase the rates of reaction beyond a certain point.
How is rate of reaction measured?
Reaction rate is calculated using the formula rate = Δ[C]/Δt, where Δ[C] is the change in product concentration during time period Δt. The rate of reaction can be observed by watching the disappearance of a reactant or the appearance of a product over time.
Does size of catalyst affect rate?
The rate of a reaction can be increased by adding a suitable catalyst. A catalyst is a substance which changes the rate of reaction but is unchanged at the end of the reaction. Only a very small amount of catalyst is needed to increase the rate of reaction between large amounts of reactants.
What is the unit for reaction rate per second?
molarity per second
The unit of measurement for a reaction rate is molarity per second (M/s).
What are KP and KC?
Ans: The difference between Kp and Kc is as follows – Kp is the equilibrium constant with respect to the atmospheric pressure and Kc is the equilibrium constant with respect to the molar concentration of the gaseous mixture.
Does adding more catalyst increase reaction rate?
Catalysts speed up chemical reactions. Only very minute quantities of the catalyst are required to produce a dramatic change in the rate of the reaction. This is really because the reaction proceeds by a different pathway when the catalyst is present. Adding extra catalyst will make absolutely no difference.
Does increasing concentration of catalyst affect rate?
Increasing the concentration of a reactant increases the frequency of collisions between reactants and will, therefore, increase the reaction rate. When a catalyst is added, the activation energy is lowered because the catalyst provides a new reaction pathway with lower activation energy.
How do u calculate rate?
Use the formula r = d/t. Your rate is 24 miles divided by 2 hours, so: r = 24 miles ÷ 2 hours = 12 miles per hour. Now let’s say you rode your bike at a rate of 10 miles per hour for 4 hours.
What is the formula to find rate?
Key Takeaways
- For a generic reaction aA+bB→C aA + bB → C with no intermediate steps in its reaction mechanism (that is, an elementary reaction), the rate is given by: r=k[A]x[B]y r = k [ A ] x [ B ] y .
- For elementary reactions, the rate equation can be derived from first principles using collision theory.
Can catalyst decrease the rate of reaction?
Catalysts can lower the activation energy and increase the reaction rate without being consumed in the reaction. Molecules joined by stronger bonds will have lower reaction rates than will molecules joined by weaker bonds, due to the increased amount of energy required to break the stronger bonds.