What are the properties of Group 8 elements?
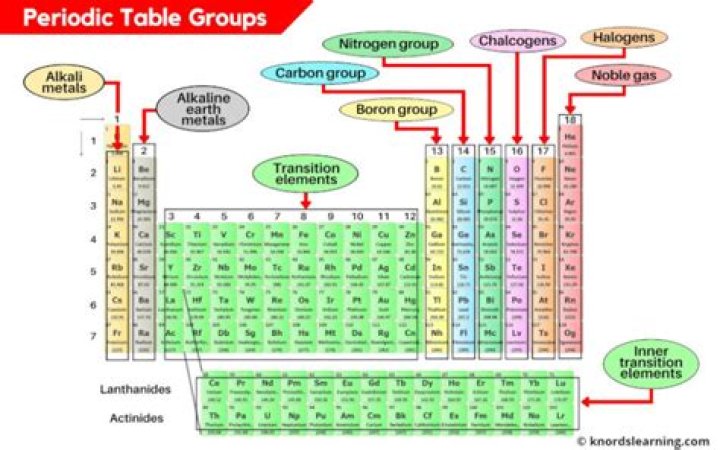
What are the properties of Group 8 elements?
Let’s take a moment to review some of the properties these nobles of the element world share:
- They are inert gases located on the right of the periodic table.
- They have a full-set of valence electrons, so they’re stable.
- They are colorless, odorless and tasteless.
- They have low melting and low boiling points.
What is the name of the Group 8 elements on the periodic table?
Group 8A (or VIIIA) of the periodic table are the noble gases or inert gases: helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and radon (Rn). The name comes from the fact that these elements are virtually unreactive towards other elements or compounds.
Is group 0 and 8 the same?
Group 0 used to be called Group 8 but this caused confusion because most elements in Group 8 have 8 electrons in their Outer Shell but Helium only has 2, so it was renamed Group 0.
What are the uses of Group 8 elements?
The noble gases are used to form inert atmospheres, typically for arc welding, to protect specimens, and to deter chemical reactions. The elements are used in lamps, such as neon lights and krypton headlamps, and in lasers. Helium is used in balloons, for deep-sea diving air tanks, and to cool superconducting magnets.
What element atomic number is 8?
Oxygen
The Elements, sorted by Atomic Number
| Atomic Number | Symbol | Name |
|---|---|---|
| 6 | C | Carbon |
| 7 | N | Nitrogen |
| 8 | O | Oxygen |
| 9 | F | Fluorine |
Why are Group 8 elements gases at room temperature?
That’s because they have eight valence electrons, which fill their outer energy level. This is the most stable arrangement of electrons, so noble gases rarely react with other elements and form compounds.
What is Group 1 called?
alkali metal
alkali metal, any of the six chemical elements that make up Group 1 (Ia) of the periodic table—namely, lithium (Li), sodium (Na), potassium (K), rubidium (Rb), cesium (Cs), and francium (Fr). The alkali metals are so called because reaction with water forms alkalies (i.e., strong bases capable of neutralizing acids).
What do the shells of Group 8 have in common?
Well, since they are the very last group on the periodic table, they have full outer shells of valence electrons. This means that on the periodic table, these are the most stable elements because the elements can only hold a certain amount of valence electrons in the outermost shell ( 8 valence electrons).
What is the element with 8 electrons?
Oxygen is the eighth element in the periodic table, with the symbol O. This means that it has eight electrons in its neutral state.
What does the 8 mean on the periodic table?
Oxygen is atomic number 8 on the periodic table. Each oxygen atom has 8 protons.
What are the 8 noble gases?
noble gas, any of the seven chemical elements that make up Group 18 (VIIIa) of the periodic table. The elements are helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), radon (Rn), and oganesson (Og).


