What are the products of SnCl4?
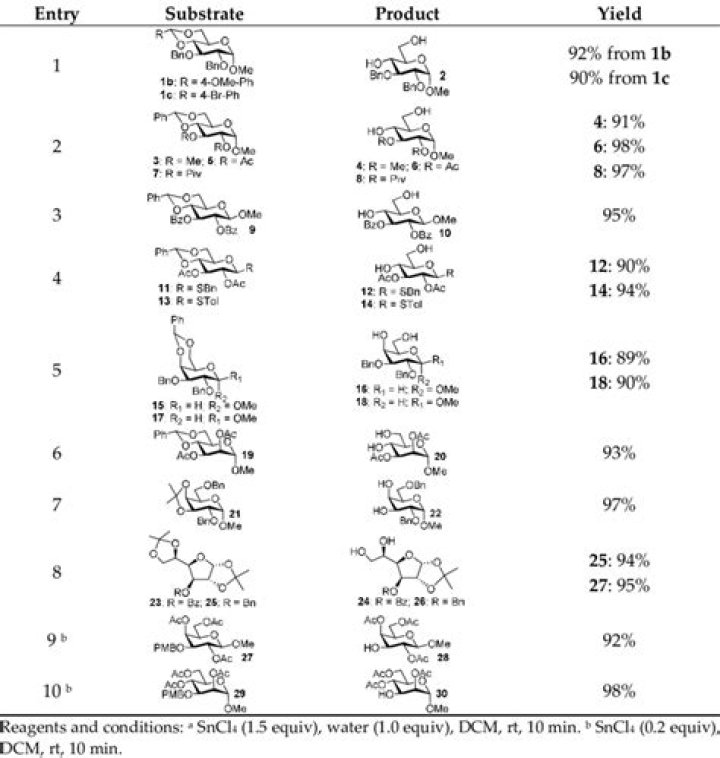
What are the products of SnCl4?
3.1Computed Properties
| Property Name | Property Value | Reference |
|---|---|---|
| Isotope Atom Count | 0 | Computed by PubChem |
| Defined Atom Stereocenter Count | 0 | Computed by PubChem |
| Undefined Atom Stereocenter Count | 0 | Computed by PubChem |
| Defined Bond Stereocenter Count | 0 | Computed by PubChem |
How is SnCl4 formed?
SnCl4 (anhydrous) can be manufactured by applying Chlorine gas to Tin metal. The formed SnCl4 (liquid) should be allowed to drip onto the Tin metal in the apparatus. Anhydrous SnCl4 will dissolve in cold water ( warm/hot will decompose it). Saturate a water solution of SnCl4 with Chlorine gas.
What is the formula for Tin IV chloride?
SnCl₄
Tin(IV) chloride/Formula
What happens when tin chloride is exposed to moist air?
Reacts violently with water and moist air. This produces corrosive hydrogen chloride (see ICSC 0163). Reacts with turpentine, alcohols and amines. This generates fire and explosion hazard.
Is SnCl4 ionic or covalent?
SnCl2 is ionic but SnCl4 is covalent.
What is the formula of tin?
Tin, ion (Sn2+)
| PubChem CID | 104883 |
|---|---|
| Structure | Find Similar Structures |
| Molecular Formula | Sn+2 |
| Synonyms | Tin(2+) Tin (II) ion tin(II) tin ion (2+) Tin, ion (Sn2+) More… |
| Molecular Weight | 118.71 |
What is Tin IV oxide formula?
SnO₂
Tin(IV) oxide/Formula
What is the formula of tin IV oxide?
Is tin a transition metal?
Tin is a chemical element with symbol Sn and atomic number 50. Classified as a post-transition metal, Tin is a solid at room temperature.
What is the oxidation number of hydrogen in H2?
0
The oxidation number for elemental hydrogen, H2, is 0.
What is the oxidation number of sns2?
+4
Tin(IV) sulfide, also known as stannic sulfide, is a chemical compound. Its chemical formula is SnS2. It has tin and sulfide ions in it. The tin is in its +4 oxidation state.