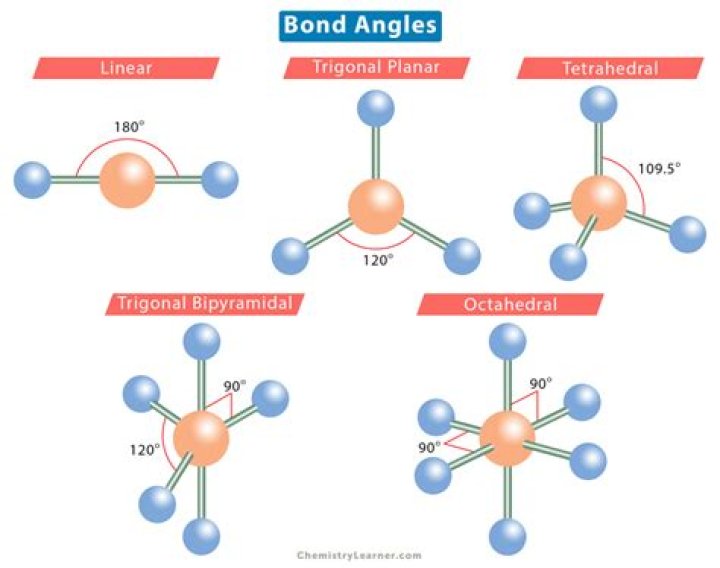
A bond angle is the geometric angle between two adjacent bonds. Some common shapes of simple molecules include: Linear: In a linear model, atoms are connected in a straight line. The bond angles are set at 180°..
Keeping this in view, what is meant by bond angle?
A bond angle is the angle between two bonds originating from the same atom in a covalent species. eg. 1: Geometrically, a bond angle is an angle between two converging lines.
Additionally, what are ideal bond angles? Bond angles also contribute to the shape of a molecule. The bond angle can help differentiate between linear, trigonal planar, tetraheral, trigonal-bipyramidal, and octahedral. The ideal bond angles are the angles that demonstrate the maximum angle where it would minimize repulsion, thus verifying the VSEPR theory.
Keeping this in view, how do bond angles work?
If the number of rods(bonds) is 2 then the shape is linear and angles are 180 degrees. If the number of rods(bonds) is 3 then the shape is triagonal planar and angles are 120 degrees. If the number of rods(bonds) is 4 then the shape is tetrahedral and angles are 109 degree 28 minutes.
What does Bond angle depend on?
Angle between two adjacent bonds at an atom in a molecule made up of three or more atoms is known as the bond angle. Generally s- character increase in the hybrid bond, the bond angle increases. (ii) Lone pair repulsion: Bond angle is affected by the presence of lone pair of electrons at the central atom.
Related Question Answers
What is Bond angle with example?
Bond angle is simply the angle between two bonds or two bonded electron pairs in a compound. For example in CH4 the bond angle is 109 degrees.What affects bond angle?
Lone pair repulsion: Bond angle is affected by the presence of lone pair of electrons at the central atom. A lone pair of electrons at the central atom always tries to repel the shared pair (bonded pair) of electrons. Due to this, the bonds are displaced slightly inside resulting in a decrease of bond angle.Which bond angle is the smallest?
| # of bonding groups/domains on 'central' atom | # of lone pair electrons on 'central' atom | Bond Angle |
| 3 | 1 | less than 109.5 |
| 2 | 2 | less than 109.5 |
| 5 | 0 | 90, 120 and 180 |
| 4 | 1 | 90, 120 and 180 |
What is the bond angle of h2o?
Water has 4 regions of electron density around the central oxygen atom (2 bonds and 2 lone pairs). These are arranged in a tetrahedral shape. The resulting molecular shape is bent with an H-O-H angle of 104.5°.How do you predict molecular shapes?
To predict the shape of a molecule: - Write the Lewis dot structure for the molecule.
- Determine the steric number of the central atom.
- Decide on the electron pair orientation based on the steric number.
- Consider the placement of lone pairs and any distortions from "regular" shapes.
What are the expected bond angles in ICl4+?
What are the expected bond angles of ICl4+? Choose all that apply: a) 90 degrees b)109.5 degrees c)120 degrees d)180 degrees.Why is so2 bent and not linear?
CO2 is linear and the SO2 structure is bent, because CO2 has a negative oxygen on each side of the positive carbon they cancel each other out. In the SO2 structure the oxygen are not in lined with each other which means there a positive and negative end.Is h2o polar or nonpolar?
A water molecule, abbreviated as H2O, is an example of a polar covalent bond. The electrons are unequally shared, with the oxygen atom spending more time with electrons than the hydrogen atoms. Since electrons spend more time with the oxygen atom, it carries a partial negative charge.Which has highest bond angle?
Also, bond angle of H 2 O < N H 3 H_{2}O<NH_{3} H2O<NH3 as lone-lone pair repulsion on oxygen is more effective compared to other repulsions (Nitrogen has only one lobe pair). Hence, ammonia has the highest bond angle.What is meant by bond enthalpy?
Bond enthalpy (also known as bond energy) is defined as the amount of energy required to break one mole of the stated bond. For example, the bond energy of a O-H single bond is 463 kJ/mol. This means that it requires 463 kJ of energy to break one mole of O-H bonds.What is bond angle strain?
Angle strain is the increase in potential energy of a molecule due to bond angles deviating from the ideal values. In the planar cyclopropane ring the internal bond angle at each carbon atom is 60º.Is h2o tetrahedral?
Water or H2O has 8 electrons around the central oxygen atom. This means there are four electron pairs arranged in a tetrahedral shape. There are two bonding pairs and two lone pairs. The resulting shape is bent with an H-O-H angle of 104.5°.Is co2 polar or nonpolar?
Carbon dioxide is non-polar because of the symmetry of its bonding. The electronegativity difference between carbon and oxygen is 1.0, which makes the bonds polar. However, the two polar bonds are at 180 degrees to each other so the dipoles cancel out.What is bond energy in biology?
Bond energy (E) is defined as the amount of energy required to break apart a mole of molecules into its component atoms.What are the bond angles for octahedral structure?
For example, methane (CH4) is a tetrahedral molecule. Octahedral: Octa- signifies eight, and -hedral relates to a face of a solid, so "octahedral" means "having eight faces". The bond angle is 90 degrees.What is the value of the bond angles in bf3?
We divide the 360 degrees of a circle by the number of electrons, i.e. 3 and get the bond angle as 120 degrees. The VSEPR theory states that when the electron-pair geometry is trigonal planar the bond angle is around 118 degrees.How many 120 bond angles are in a hydrocarbon?
Answer and Explanation: Carbon atoms with four single bonds have bond angles of approximately 109.5 degrees; C atoms with two single bonds and one double bond, 120 degrees;Do double bonds affect bond angle?
Multiple bonds contain a higher electronic-charge density than do single bonds, so multiple bonds also represent larger electron domains ("fatter balloons"). However, the double bond seems to act much like a nonbonding pair of electrons, reducing the ClCCl bond angle from 120° to 111°.What bond angle is sp2?
An sp2 hybridized atomic orbital is the mixing of 1 S orbital and 2 P orbitals, a sp2 hybridized atom has 3 sigma bonds so its molecular geometry is trigonal with 120 degree bond angles.