What are lactones properties?
What are lactones properties?
Lactones are cyclic esters. Many simple examples occur in essential oils, as well as more complex molecules, which have low volatility. Sesquiterpene lactones are notorious for their tendency to be skin sensitizers (Warshaw & Zug 1996).
How do you synthesize lactones?
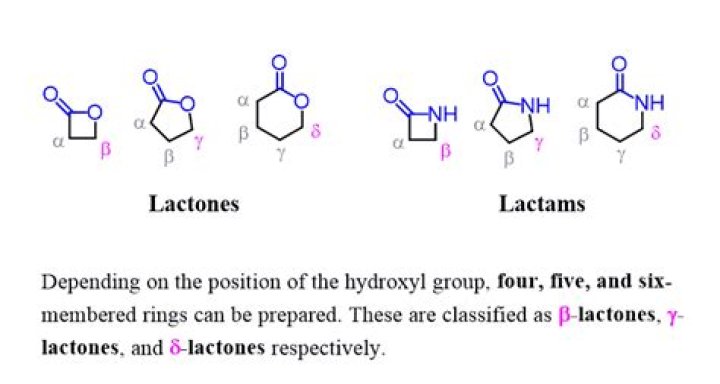
Lactones are formed by intramolecular esterification of the corresponding hydroxycarboxylic acids, which takes place spontaneously when the ring that is formed is five- or six-membered.
What are lactones and by what chemical reaction are they produced?
lactone, any of a class of cyclic organic esters, usually formed by reaction of a carboxylic acid group with a hydroxyl group or halogen atom present in the same molecule.
How do you identify lactone?
Cyclic esters are called lactones. In these cases the COOH and OH groups that combine to form water are part of the same molecule (see above Classes of carboxylic acids: Hydroxy and keto acids). Lactones are known with rings of all sizes from 3 to 20 or more, although 3-membered rings are extremely unstable.
What are lactones examples?
Some Examples of Lactones are Macrolides, Ellagic acid, Kavalactones, Lactide, Valoneic, Tergallic acid diactone.
What is the functional group of lactones?
A lactone is an ester in which the functional group of the ester has become part of a ring structure with carbon atoms.
What does lactones smell like?
The scent of lactones is often defined as creamy and the word itself partly hints at its meaning (Lat. lac, lactis – milk). Isoamylacetate basically smells like pears and bananas, while butyl isobutyrate is a veritable chaos of tropical fruit and overripe strawberries.
How are lactones used to make carbohydrates?
The synthesis and uses of different kinds of carbohydrate-based lactones are described. This group of compounds includes aldonolactones, other related monocyclic lactones and bicyclic systems. The latter can arise from uronic acids, carboxymethyl ethers or glycosides, or from C-branched sugars.
Where are lactones found?
Lactones are mainly found in expressed oils and some absolutes like jasmine (Clarke 2002). They are cyclic esters derived from lactic acid (Baser & Demirci 2007) and have an oxygen atom double bonded to a carbon atom. The carbon atom is attached to another oxygen atom that is part of a closed ring (Table 3-16).
Are lactones esters?
Lactones are cyclic esters of organic acids. It is a condensation product of an alcohol group and a carboxylic acid group in the same molecule of hydroxycarbonic acid.
Is Lactose a lactone?
As nouns the difference between lactose and lactone is that lactose is while lactone is (organic chemistry) a cyclic intramolecular ester derived from a hydroxy acid.
What is Lactonization reaction?
The nucleophile-catalyzed aldol lactonization (NCAL) reaction of carboxyl-derived zwitterions and unactivated aldehydes has been applied to the catalytic asymmetric synthesis of bicyclic β-lactones (Equation 31) <2005JOC2835>. A review of the methodology for β-lactone synthesis has been published <1999T6403>.


