The ions or molecules that bind to transition-metal ions to form these complexes are called ligands (from Latin, "to tie or bind"). Although coordination complexes are particularly important in the chemistry of the transition metals, some main group elements also form complexes..
Furthermore, what are coordination complexes used for?
Coordination compounds include such substances as vitamin B12, hemoglobin, and chlorophyll, dyes and pigments, and catalysts used in preparing organic substances. A major application of coordination compounds is their use as catalysts, which serve to alter the rate of chemical reactions.
Similarly, how do you identify ligands in complex ions? Writing the (Line) Formula of a Complex:
- Identify the central metal ion.
- Identify the oxidation state on the central metal ion (shown in Roman numerals parantheses)
- Identify the ligands.
- Identify the number of ligands.
- Calculate the total charge on the ligands.
- Calculate the charge on the complex ion.
Also question is, what are the types of ligands?
Types of Ligands
- Unidentate ligands: Ligands with only one donor atom, e.g. NH3, Cl-, F- etc.
- Bidentate ligands: Ligands with two donor atoms, e.g. ethylenediamine, C2O42-(oxalate ion) etc.
- Tridentate ligands: Ligands which have three donor atoms per ligand, e.g. (dien) diethyl triamine.
What are complexes in chemistry?
Complex, in chemistry, a substance, either an ion or an electrically neutral molecule, formed by the union of simpler substances (as compounds or ions) and held together by forces that are chemical (i.e., dependent on specific properties of particular atomic structures) rather than physical.
Related Question Answers
How complexes are formed?
A complex ion is a species formed between a central metal ion and one or more surrounding ligands, molecules or ions that contain at least one lone pair of electrons. The formation of a complex ion by adding a complexing agent increases the solubility of a compound.What is meant by coordination complex?
A coordination complex is the product of a Lewis acid-base reaction in which neutral molecules or anions (called ligands) bond to a central metal atom (or ion) by coordinate covalent bonds. Ligands are Lewis bases - they contain at least one pair of electrons to donate to a metal atom/ion.What is a coordination reaction?
coordination: The reaction of one or more ligands with a metal ion to form a coordination compound. redox: A reversible chemical reaction in which one reaction is an oxidation and the reverse is a reduction. donor atom: The atom within a ligand that is bonded to the central atom or ion within a coordination complex.What is a complexation reaction?
complexation reaction. [‚käm·plek′sā·sh?n rē‚ak·sh?n] (chemistry) A chemical reaction that takes place between a metal ion and a molecular or ionic entity known as a ligand that contains at least one atom with an unshared pair of electrons.How are ligands formed?
Ligands are ions or neutral molecules that bond to a central metal atom or ion. Ligands act as Lewis bases (electron pair donors), and the central atom acts as a Lewis acid (electron pair acceptor). Ligands have at least one donor atom with an electron pair used to form covalent bonds with the central atom.What do ligands of coordination complexes have in common?
According to this model, transition-metal ions form coordination complexes because they have empty valence-shell orbitals that can accept pairs of electrons from a Lewis base. Ligands must therefore be Lewis bases: They must contain at least one pair of nonbonding electrons that can be donated to a metal ion.Why are metal complexes important?
One of the most important properties of metallic elements is their ability to act as Lewis acids that form complexes with a variety of Lewis bases. Second, many industrial catalysts are metal complexes, and such catalysts are steadily becoming more important as a way to control reactivity.How is EAN calculated?
Generally EAN of central metal ion will be equal to the number of electrons in the nearest noble gas. If the EAN of the central metal is equal to the number of electrons in the nearest noble gas then the complex possess greater stability. EAN= [Z metal – (ox. state of the metal) + 2(coordination number of the metal)].What are ligands with examples?
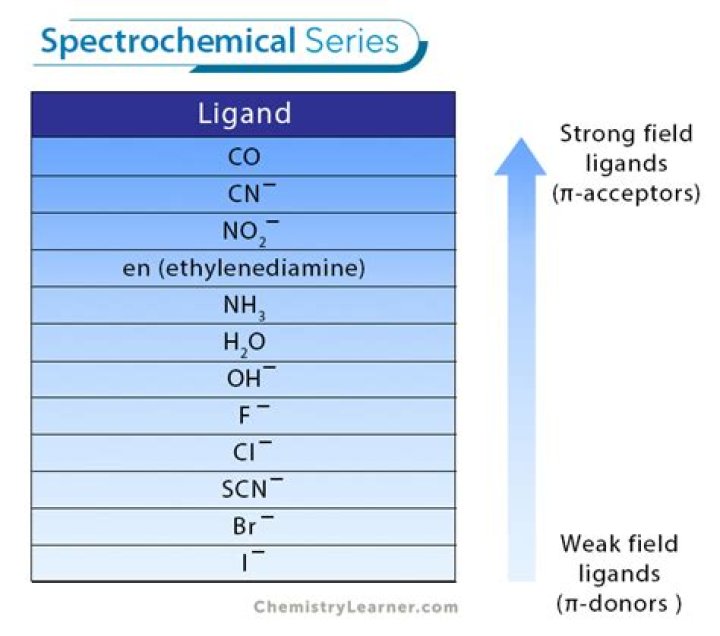
Examples of common ligands are the neutral molecules water (H2O), ammonia (NH3), and carbon monoxide (CO) and the anions cyanide (CN-), chloride (Cl-), and hydroxide (OH-). Occasionally, ligands can be cations (e.g., NO+, N2H5+) and electron-pair acceptors.What type of ligand is EDTA?
EDTA is a hexadentate ligand, which means that it binds six times. It binds twice at the nitrogens and four at the oxygens. EDTA is used most commonly as salts and in a dry form. EDTA is a great chelating agent, allowing multiple bindings in a coordination complex.How do you classify ligands?
Ligands can be classified on the basis of many things. The most common classification of ligands is on the basis of their binding sites with the central metal atom or ion. On the basis of the number of sites, ligands can be classified as monodentate, bidentate, polydentate etc. ligands.What are positive ligands?
Are there positively charged ligands which can bind to a central metal atom to form coordination compounds? I know that ligands are Lewis bases which donate a pair of electrons, and the central metal atom is usually a Lewis acid. However, a positive charge on a species usually means it is electron-deficient.What type of ligand is CN?
This type of ligand, capable of sharing 1 pair of electrons, is referred to as a monodentate ligand. Ethylenediamine (1,2 diaminoethane) has two lone pair of electrons that may be shared with a metal atom or ions. Common ambidentate ligands include cyanide (CN-), nitrite (NO2 -), thiocyanate (SCN-).What are natural ligands?
In biochemistry, a ligand is any molecule or atom which binds reversibly to a protein. This is because the key properties of a ligand are found in its chemical structure. If that structure can be recreated in the laboratory, the synthetic ligand will be able to interact in the same ways a natural ligand acts.What are chelates with example?
A chelate is a chemical compound composed of a metal ion and a chelating agent. A chelating agent is a substance whose molecules can form several bonds to a single metal ion. In other words, a chelating agent is a multidentate ligand. An example of a simple chelating agent is ethylenediamine.What type of ligand is water?
The molecules or ions surrounding the central metal ion are called ligands. Simple ligands include water, ammonia and chloride ions. What all these have got in common is active lone pairs of electrons in the outer energy level. These are used to form co-ordinate bonds with the metal ion.Is a ligand a protein?
A ligand is a small molecule that is able to bind to proteins by weak interactions such as ionic bonds, hydrogen bonds, Van der Waals interactions, and hydrophobic effects. In some cases, a ligand also serves as a signal triggering molecule. For example, oxygen is the ligand that binds to both hemoglobin and myoglobin.What are examples of complex ions?
Naming complex ions For example: F- fluoro, CN- cyano, Cl- chloro, OH- hydroxo (H2O is aqua, NH3 is ammine).Which is strongest ligand?
CO