What are 5 examples of ionic bonds?
What are 5 examples of ionic bonds?
Some ionic bond examples include:
- NaCl: sodium chloride.
- NaBr: sodium bromide.
- NaF: sodium fluoride.
- NaI: sodium iodide.
- KF: potassium fluoride.
- KCl: potassium chloride.
- KI: potassium iodide.
- KBr: potassium bromide.
What is a common example of ionic bonding?
Examples of ionic compounds in everyday life include table salt, baking soda, lye, Epsom salt, and bleach. There are many examples of ionic compounds in everyday life. Ionic compounds consist of atoms joined together by ionic bonds. Many ionic compounds are binary compounds formed by a metal and a nonmetal.
What are the example of ionic?
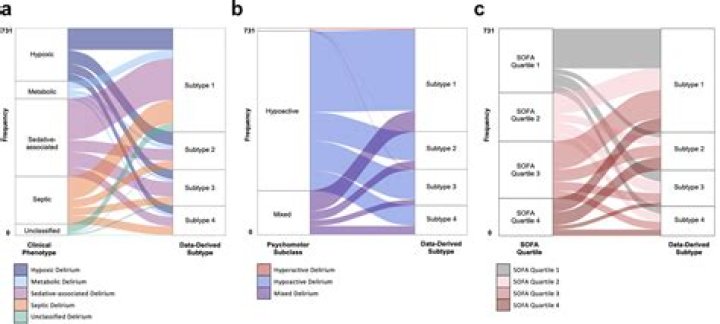
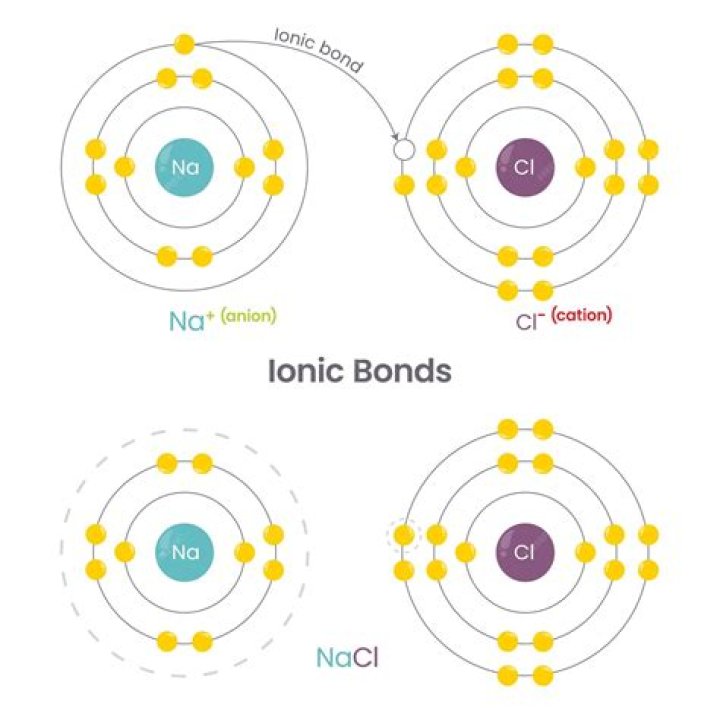
Table salt is an example of an ionic compound. Sodium and chlorine ions come together to form sodium chloride, or NaCl. The sodium atom in this compound loses an electron to become Na+, while the chlorine atom gains an electron to become Cl-.
What are example of ionic?
Ionic Compounds Are Balanced Table salt is an example of an ionic compound. Sodium and chlorine ions come together to form sodium chloride, or NaCl. The sodium atom in this compound loses an electron to become Na+, while the chlorine atom gains an electron to become Cl-.
What are examples of covalent bonds found in your everyday life?
10 Covalent Bond Examples in Real Life
- Water.
- Sugar.
- Oxygen.
- Carbon Dioxide.
- LPG.
- Vinegar.
- Nail Polish Remover.
- Diamonds.
How do you write ionic bonds?
To find the formula of an ionic compound, first identify the cation and write down its symbol and charge. Then, identify the anion and write down its symbol and charge. Finally, combine the two ions to form an electrically neutral compound.
Is nal molecular or ionic?
Sodium iodide is a metal iodide salt with a Na(+) counterion. It is an inorganic sodium salt and an iodide salt. Sodium iodide is a water-soluble ionic compound with a crystal lattice.
Is CH4 ionic compounds?
Each ball in the gas diagram above represents one methane molecule. Is CH4 ionic or covalent? Methane, CH4, is a covalent compound with exactly 5 atoms that are linked by covalent bonds. We draw this covalent bonding as a Lewis structure (see diagram).
How do we use ionic bonding in everyday life?
Let’s discuss a few examples of ionic bonding in daily life.
- Table Salt.
- Iodized Salt.
- Fluoride in Toothpaste.
- Baking Soda.
- Washing Soda.
- Household Bleach.
- Preservative.
- Anti-caking Agent.
What are examples of ionic and covalent bonds?
Ionic vs Covalent Bond Summary
| Ionic Bonds | Covalent Bonds | |
|---|---|---|
| Boiling Point | High | Low |
| State at Room Temperature | Solid | Liquid or Gas |
| Examples | Sodium chloride (NaCl), Sulfuric Acid (H2SO4 ) | Methane (CH4), Hydrochloric acid (HCl) |
| Chemical Species | Metal and nometal (remember hydrogen can act either way) | Two nonmetals |