Is trans-2-butene symmetrical?
Is trans-2-butene symmetrical?
Trans-2-butene has a symmetrical structure. This is because when symmetry elements are considered , trans-2-butene has an an axis of symmetry, a plane of symmetry, and a center of symmetry compared to cis-2-butene . This makes trans-2-butene more symmetrical.
How is trans alkene symmetrical?
Trans alkenes have a C2h symmetry, identified as such because it has a C2 rotational axis (you need to rotate 180° to have an identical molecule) and a mirror plane perpendicular to that axis σh. You need to follow the symmetry flowchart to assign symmetry groups.
Do trans isomers have symmetry?
Trans isomers, on the other hand, are more linear. Trans alkenes, therefore, are more symmetrical and hence less polar, have lower boiling points and higher melting points. Cis alkenes, on the other hand, are generally less symmetrical, and hence more polar, have higher boiling points and lower melting points.
Is trans more symmetrical than cis?
Due to this boiling point of cis isomers are greater than trans isomers. Trans isomers have higher melting point than Cis isomers as the former are more symmetrical than the latter. More symmetrical a molecule more easily it can fit well in to the crystal lattice, hence higher melting point.
Does CIS butene have plane of symmetry?
It’s a planar molecule. So it has plane of symmetry.
What is plane of symmetry in organic chemistry?
A plane of symmetry is an imaginary plane that bisects a molecule into halves that are mirror images of each other. In 2, the horizontal plane that bisects all six atoms in the molecule bisects the molecule into halves that are mirror images of each other. Therefore, it is a plane of symmetry. eg.
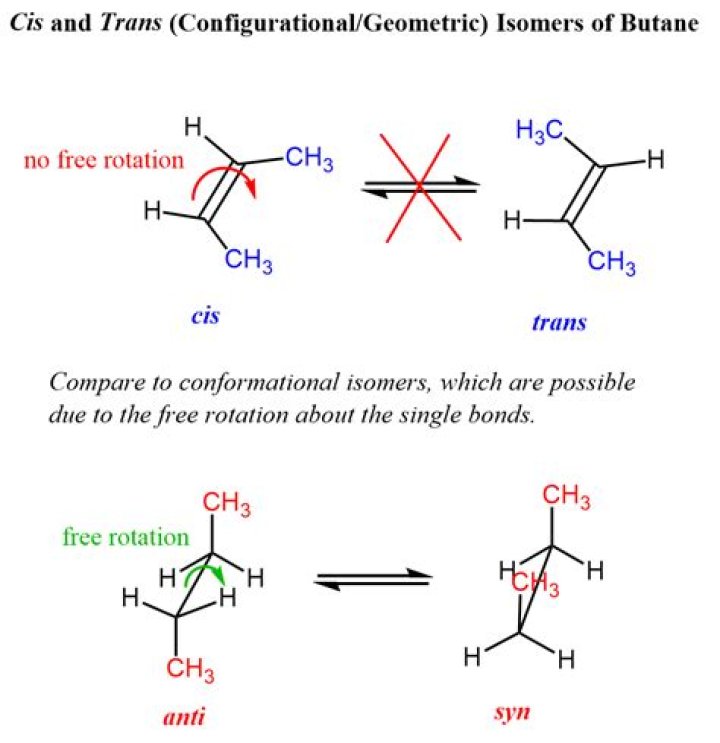
What is cis trans isomerism in alkenes?
Cis-trans isomers are compounds that have different configurations (groups permanently in different places in space) because of the presence of a rigid structure in their molecule. Alkenes and cyclic compounds can exhibit cis-trans isomerism.
How do you find cis trans isomers?
Consider the longest chain containing the double bond: If two groups (attached to the carbons of the double bond) are on the same side of the double bond, the isomer is a cis alkene. If the two groups lie on opposite sides of the double bond, the isomer is a trans alkene.
Is 2 butene a cis trans isomer?
2-Butene is an acyclic alkene with four carbon atoms. It is the simplest alkene exhibiting cis/trans-isomerism (also known as (E/Z)-isomerism); that is, it exists as two geometric isomers cis-2-butene ((Z)-2-butene) and trans-2-butene ((E)-2-butene).
Is trans symmetrical?
In a trans isomer, the dipoles of the substituents will cancel out due to being on opposite sides of the molecule. As a general trend, trans alkenes tend to have higher melting points and lower solubility in inert solvents, as trans alkenes, in general, are more symmetrical than cis alkenes.
What is benzene Point Group?
Benzene contains a main C6 axis which contains S6 and S3 axes. Perpendicular to the C6 axis are 6 C2 axes. There is a σh, 3 σv and 3 σd planes, as well as an inversion centre. Hence Benzene belongs to the D6h point group.


