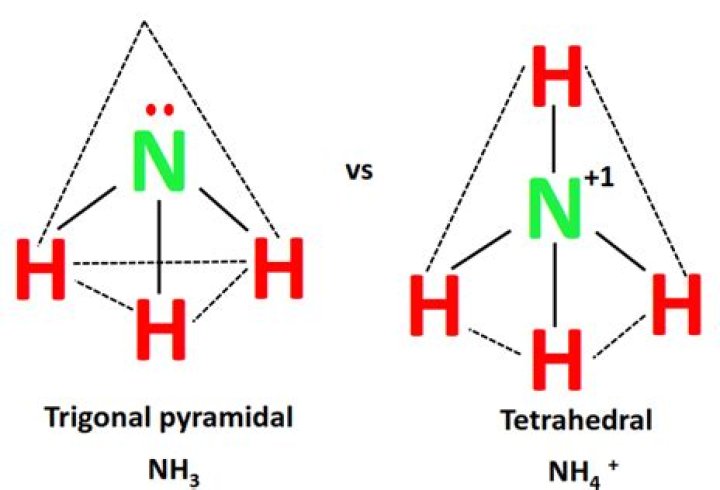
NH3 (Ammonia) electron geometry is“Tetrahedral” but its molecular geometry is“Trigonal Pyramidal”. the H atoms are being pushed downby the electron pair on top and have an approximate bond angle of109.5 degree..
Considering this, is nh3 tetrahedral or trigonal pyramidal?
For example; four electron pairs are distributed in atetrahedral shape. If these are all bond pairs the moleculargeometry is tetrahedral (e.g. CH4). If there is one lonepair of electrons and three bond pairs the resulting moleculargeometry is trigonal pyramidal (e.g.NH3).
is ammonia a tetrahedral? The ammonia molecule contains three single bondsand one lone pair on the central nitrogen atom (see Figure 8). Thedomain geometry for a molecule with four electron pairs istetrahedral, as was seen with CH 4 . Recall thatthe bond angle in the tetrahedral CH 4 moleculeis 109.5°.
Accordingly, what is the electron geometry of nh3?
NH3 electron geometry is: 'Tetrahedral,' as ithas four group of electrons. One group has an unshared pairof electrons. 'N' has tetrahedral electronicgeometry. Thus, Ammonia is an example of the molecule inwhich the central atom has shared as well as an unshared pair ofelectrons.
Why is the shape of ammonia not tetrahedral?
An example of trigonal pyramid molecular geometrythat results from tetrahedral electron pair geometryis NH3. The molecule is three dimensional as opposed to theboron hydride case which was a flat trigonal planarmolecular geometry because it did not have a loneelectron pair.
Related Question Answers
Is ammonia a planar?
Ammonia (NH3) is not trigonalplanar, however. It is trigonal pyramidal because it isattached to four "things": the three hydrogens and a non-bondingpair of electrons (to fulfill nitrogen's octet).How many lone pairs are in Hf?
three lone pairs
Is ammonia a trigonal pyramidal?
Trigonal pyramidal geometry inammonia The nitrogen in ammonia has 5 valence electronsand bonds with three hydrogen atoms to complete the octet. Thiswould result in the geometry of a regular tetrahedron with eachbond angle equal to cos−1(−13)≈ 109.5°.Why is ammonia a trigonal pyramidal?
…The ammonia molecule has a trigonalpyramidal shape with the three hydrogen atoms and an unsharedpair of electrons attached to the nitrogen atom. It is a polarmolecule and is highly associated because of strong intermolecularhydrogen bonding. What is the bond angle of h2o?
Water has 8 electrons around the central oxygen atom.This means there are four electron pairs arranged in a tetrahedralshape. There are two bonding pairs and two lone pairs. Theresulting shape is bent with an H-O-H angle of104.5°.How do you determine polarity?
Step 2: Identify each bond as either polaror nonpolar. (If the difference in electronegativity for the atomsin a bond is greater than 0.4, we consider the bond polar.If the difference in electronegativity is less than 0.4, the bondis essentially nonpolar.) If there are no polar bonds, themolecule is nonpolar.Is nh3 linear?
The linear combination of atomic orbitals isdrawn and detailed in Figure 4. Ammonia or NH3 has 8 valenceelectrons, consisting of a lone pair on its nitrogen and 3 N-Hsigma bonds.Does nh3 violate the octet rule?
Nitrogen, the next nonmetal, has 5 electrons in thevalence shell, so it needs to combine with 3 hydrogen atoms tofulfill the octet rule and form a stable compound calledammonia (NH3). In the ammonia molecule, these electrons arepaired and unshared, meaning that they are not engaged inbonding.Is h2o polar or nonpolar?
A water molecule, abbreviated as H2O, is anexample of a polar covalent bond. The electrons areunequally shared, with the oxygen atom spending more time withelectrons than the hydrogen atoms. Since electrons spend more timewith the oxygen atom, it carries a partial negativecharge.Is ammonia polar or nonpolar?
Ammonia is a polar molecule: See graphic on the left. The trigonal pyramid geometrywith the one lone electron pair contributes to the effect. Theelectrostatic potential clearly shows that the nitrogen ispartially negative whereas the hydrogens are partiallypositive.Is XeF4 planar?
The VSEPR structure of XeF4 is squareplanar. It is an octahedral but because of the lone pairs,it dictate there 6 Domains around the central atom and the VSEPRtheory states any AX4E2 specie with 2 lone pairs is squareplanar.Is h20 planar?
Is water a planar molecule? - Quora. Yes, wateris a planer molecule. But due to presence of lone pair on O thereexist lone pair-lone pair and lone pair -bond pair repulsion due towhich the shape of water get destroyed and resulting shape isdistorted tetrahedral or V- shape or angular shape.What is the geometry formula for ammonia?
NH3 (Ammonia) electron geometry is“Tetrahedral” but its molecular geometry is“Trigonal Pyramidal”.What are repelled in the Vsepr theory?
Valence Shell Electron Pair Repulsion Theory(VSEPR). Say it five times quickly. VSEPR theorysimply states that the pairs of electrons in a chemical compoundrepel each other and move as far from each other as possiblebecause they have the same charge.How do you know if a molecule is tetrahedral?
The four bond pairs are arranged about the C atom,pointing toward the corners of a regular tetrahedron. Thisshape minimizes the repulsion between the bond pairs. The109.5° angle is the same for all H-C-H bond angles and iscalled the tetrahedral bond angle. The shape of the CH4molecule is tetrahedral.What is ammonia made of and what are some of its uses?
Ammonia is made out of one nitrogen andthree hydrogen atoms. Its structure is tetrahedral.Ammonia is used in nitric acid production, as a fertilizer,and a cleaning solution. NH3, normally found as a gas, it iscaustic and harmful in longterm exposure.Does ammonia have a 2p orbital?
In the ammonia molecule (NH3), 2s and2p orbitals create four sp3hybridorbitals, one of which is occupied by a lone pair ofelectrons.