Is dense phase supercritical?
Is dense phase supercritical?
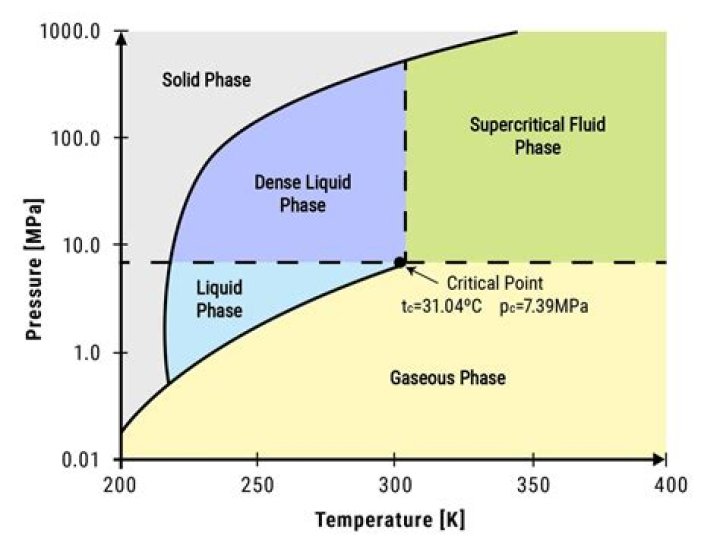
temperature and critical pressure is labeled as “supercritical region”, while the region above the critical pressure, but below the critical temperature, is called the “dense phase region”[11-13].
What state is a supercritical fluid in?
chemical separation Gaseous substances beyond a specific temperature and pressure (the critical point) become a supercritical fluid, a state that is more dense than a gas but less dense than a liquid. A supercritical fluid can thus dissolve (i.e., solvate) species better than a gas…
Is co2 a compressible fluid?
When a pure compound, in gaseous or liquid state, is heated and compressed above the critical temperature and pressure, it becomes a dense, highly compressible fluid that demonstrates properties of both liquid and gas. …
What is super critical temperature?
The supercritical condition of a steam-water cycle is a state at which its temperature and pressure are above its thermodynamic critical point, where the pressure of the steam water is 22.12 MPa, the temperature is 647.14 K, and the density is 324 kg/m3.
What is the density of supercritical CO2?
0.4 to 0.6 g cm-3
The range of the underground supercritical CO2 density in the study area was 0.4 to 0.6 g cm-3.
How is supercritical CO2 made?
It is a solid phase of CO2 when frozen. At standard temperature and pressure (STP), CO2 usually behaves as a gas. If dry ice is put in an enclosed vessel, it will sublimate to become a gas and the pressure will increase depending on the mass of dry ice until the desired pressure (supercritical pressure) is achieved.
Is supercritical CO2 a liquid?
Supercritical carbon dioxide (sCO. 2 ) is a fluid state of carbon dioxide where it is held at or above its critical temperature and critical pressure. Carbon dioxide usually behaves as a gas in air at standard temperature and pressure (STP), or as a solid called dry ice when cooled and/or pressurised sufficiently.
What is dry ice made of?
Dry ice has just one ingredient: carbon dioxide. Technicians create dry ice by pumping liquid carbon dioxide into holding tanks, which reduces the temperature to -109° F and pressurizes the substance into solid blocks or pellets.
Why dry ice is used for refrigeration of food?
Dry ice is mostly used for refrigeration purposes. It is often used when transporting non-frozen foods and drinks from one state or country to another. This is more practical than normal ice because it is colder, last longer, and melts into a gas instead of a liquid, leaving minimal cleanup.
Which gas has highest density?
The more dense carbon dioxide gas has filled the container. Natural gas, methane, is less dense than the carbon dioxide, so it floats to the top of the more dense carbon dioxide….
| Densities of Common Elements and Compounds | |
|---|---|
| Substance | Density grams per mL |
| Hydrogen gas | 0.000089 |
| Helium gas | 0.00018 |
| Air | 0.00128 |
What is subcritical for boiling water?
Subcritical water is liquid water under pressure at temperatures above usual boiling point, 100 °C (212 °F). It is also known as “subcritical water” or “pressurized hot water.” At subcritical state, water is maintained in liquid form by apply pressure.
Are gasses dense?
And the answer is yes. A gas can be more dense than a liquid. The density of a gas is dependent upon the pressure and the temperature that it is under. The density of a gas is directly proportional to the pressure and indirectly proportional to the temperature.
What is the dense phase?
The dense phase is often referred to as a “dense fluid” to distinguish it from normal gas and liquid (see Figure 1). Dense phase is a fourth (Solid, Liquid, Gas, Dense) phase that cannot be described by the senses. The word “fluid” refers to anything that will flow and applies equally well to gas and liquid.
What is dense phase transportation of natural gas?
Transportation of Natural Gas in Dense Phase. When a pure compound, in gaseous or liquid state, is heated and compressed above the critical temperature and pressure, it becomes a dense, highly compressed fluid that demonstrates properties of both liquid and gas.
What is a dense phase pneumatic conveying system?
WHAT IS A DENSE PHASE PNEUMATIC CONVEYING SYSTEM? Smoot’s Dense Phase Pneumatic Conveying System is a high-pressure, low-velocity system that moves abrasive or easily degradable dry materials down the line gently and efficiently.
How does density affect the viscosity of a gas phase?
The higher density at higher pressure in the dense phase allows transporting more mass per unit volume, resulting in higher CAPEX. However, the OPEX reduction usually offsets the CAPEX increment. As shown in the following sections, the value of the dense phase viscosity is very similar to gas phase viscosity.