Carbon disulfide, also known as carbonbisulfide, is a chemical compound. It consists ofcarbon and sulfide ions. It contains carbon inits +4 oxidation state and sulfur in its -2 oxidationstate..
Regarding this, what kind of compound is carbon disulfide?
Carbon disulfide (CS2), also calledCarbon Bisulfide, a colourless, toxic, highly volatile andflammable liquid chemical compound, large amounts of whichare used in the manufacture of viscose rayon, cellophane, andcarbon tetrachloride; smaller quantities are employed insolvent extraction processes or converted into
Furthermore, what is carbon disulfide made of? Commercial carbon disulfide is made bycombining carbon and sulfur at very high temperatures.Several industries use carbon disulfide as a raw material tomake such things as rayon, cellophane, and carbontetrachloride. Currently, the largest user of this chemical is theviscose rayon industry.
Correspondingly, is carbon disulfide an organic compound?
Carbon disulfide. Description: Carbondisulfide is an organosulfur compound and aone-carbon compound.
What is the chemical name of cs2?
Carbon disulfide
Related Question Answers
Is cs2 miscible in water?
Can CS2 dissolve in water, why or why not?Cs2 is non polar molecule hence it is inslouble inwater .What type of bond is cs2?
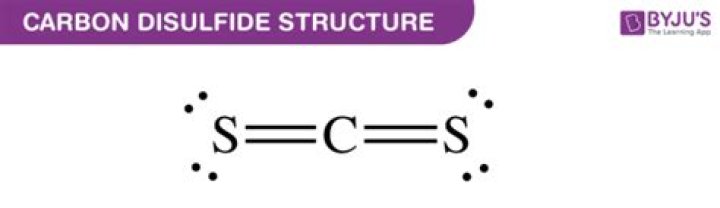
As the hybridization of CS2 is sp hybridization,the Carbon atom is in center bonding with two sulfur atomsforms the bond angle of 180 degrees, making the moleculargeometry of CS2 molecule linear.Why is carbon disulfide linear?
Is Carbon Disulfide Polar? CS2 is a non-polarmolecule because it has a linear geometric shape with asymmetric distribution of charge. Simply put, there exists a180° angle between the C-S bonds. This leads to thecancellation of the dipole moments arising due to each of thebonds.Is cs2 polar or nonpolar?
Carbon disulfide is a non-polar molecule.Polarity in relatively simple terms arises as a result ofuneven sharing of bond electrons in a covalent molecule. The causeof this is differences in electronegativities of the elementsmaking up the molecule.What is the formula of carbon disulfide?
CS2
How do you name compounds?
Rules for Naming Molecular Compounds: - Remove the ending of the second element, and add“ide” just like in ionic compounds.
- When naming molecular compounds prefixes are used to dictatethe number of a given element present in the compound.
- If there is only one of the first element, you can drop theprefix.
How is cs2 formed?
The five mechanisms of CO2 formation are asfollows: Chemical Interaction of CS2 and hydrogen sulfide(H2S) present in natural gas at high temperatures, resulting in CO2formation;Combustion of CS2 in the presence of oxygenproducing SO2 and CO2;Photolysis of CS2 leading to theformation of COS, CO, and SO2, which areWhat is ccl4 in chemistry?
Wikipedia. License. Carbon tetrachloride, also known bymany other names (the most notable being tetrachloromethane, alsorecognised by the IUPAC, carbon tet in the cleaning industry,Halon-104 in firefighting, and Refrigerant-10 in HVACR) is anorganic compound with the chemical formulaCCl4.What is carbon disulfide made up of?
Carbon disulfide is made up of molecules.Although the molecules are composed of atoms, it is notcorrect to say carbon disulfide is made of atoms.Remember, a molecule has properties that are unique. Thus, no atomhas the properties of carbon disulfide.Is cs2 an organic compound?
CS2 doesn't have any C-C or C-H bonds whichdefine the characteristics of many organic reactions. CO2 isinorganic in the same way. Urea, H2NCONH2, is considered anorganic compound but I think partly for historical reasons;it has neither C-H nor C-C bonds. It's unreactive with respect tomost organic reactions.What is the name of p4s3?
Phosphorus sesquisulfide EINECS:215-245-0. Phosphorous sesquisulfide. Phosphorussesquisulfide [Wiki] Phosphorus sulfide(P4S3)Is carbon a metal?
Carbon is a solid non-metal. It cannot becalled a metal, since it doesn't possess any physicalcharacteristics of one. It's not lustrous, it is not malleable, itdoes not conduct electricity (however, it's allotrope, graphite candue to vast electron de-localization within carbonlayers.What is the correct chemical name for a compound with the formula n2o3?
Dinitrogen trioxide is the chemical compound with theformula N2O3.Why is cs2 a liquid at room temperature?
CO2 and CS2 can exist in all 3 phases atdifferent temperatures and pressures. They have differentspecific properties because they are different molecules. Atroom T CS2 is a very low boiling liquid CO2under pressure is also a liquid but with a higher vaporpressure.What is a molecular compound?
A molecular compound is formed when two or moreatoms join together chemically through covalent bonds. Anycompound will be considered as molecule that containsat least two different elements. All compounds aremolecules but not all molecules arecompounds.