At room temperature, ammonia is a colorless, highly irritating gas with a pungent, suffocating odor. In pure form, it is known as anhydrous ammonia and is hygroscopic (readily absorbs moisture). Ammonia has alkaline properties and is corrosive. Ammonia gas is easily compressed and forms a clear liquid under pressure..
Subsequently, one may also ask, is nh4 a gas?
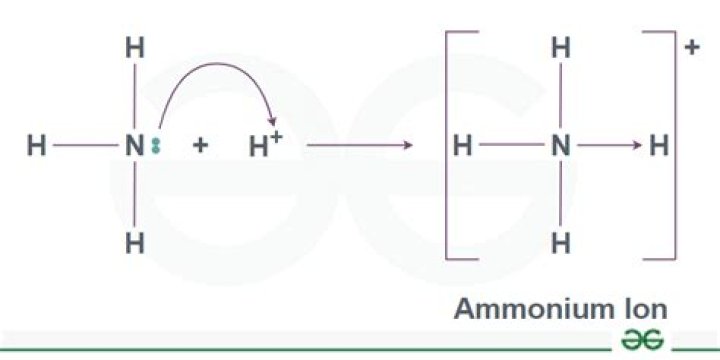
NH3 (ammonia) is a gas and sometimes called toxic or free ammonia. This type of ammonia is the dangerous part. NH4 (ammonium) is a nontoxic salt. Under normal conditions, NH3 (ammonia) and NH4 (ammonium) will both be present in aquarium water.
Similarly, is Ammonium the same as ammonia? Ammonia contains one Nitrogen and three Hydrogen whereas Ammonium contains one Nitrogen and Four Hydrogen. Ammonia is a weak base and is un-ionized. On the other hand, Ammonium is ionised. One of the noticeable differences between the two is that Ammonia gives out a strong smell whereas Ammonium does not smell at all.
Keeping this in view, is ammonium a solid?
Formation of ammonium compounds can also occur in the vapor phase; for example, when ammonia vapor comes in contact with hydrogen chloride vapor, a white cloud of ammonium chloride forms, which eventually settles out as a solid in a thin white layer on surfaces.
What is ammonium used for?
Uses and Benefits Ammonium hydroxide is used as a cleaning agent and sanitizer in many household and industrial cleaners. Ammonium hydroxide is also used in the manufacture of products such as fertilizer, plastic, rayon and rubber.
Related Question Answers
Is ammonia an acid or base?
Ammonia is normally a base, but in some reactions it can act like an acid. Ammonia acts as a base. It accepts a proton to form ammonium. Ammonia also acts as an acid.Is ammonia an ion?
Description: Inorganic compounds that include a positively charged tetrahedral nitrogen (ammonium ion) as part of their structure. The ammonium (more obscurely: aminium) cation is a positively charged polyatomic cation with the chemical formula NH4+. It is formed by the protonation of ammonia (NH3).Is nh4 solid liquid or gas?
In the soil, ammonia gas (NH3) is not stable; it can convert to ammonium (NH4), a solid, or be lost as a gas.What is total ammonia?
Total Ammonia in Water and Wastewater. In water or wastewater disinfection applications, Total Ammonia (expressed as NH3-N) is the sum of the free ammonia-nitrogen plus the amount of nitrogen from ammonia that has combined with chlorine.What is ammonia toxicity?
[6] Ammonia toxicity occurs when the ammonia content in the blood supersedes the liver's capacity to eliminate it; this could be a result of either overproduction such as in congenital hyperammonemia or under-elimination such as in liver cirrhosis. Inhalation of anhydrous ammonia gas or vapors of liquid ammonia.Is nh4 stable?
The neutral ammonium molecule (NH4) is described by a wavefunction composed of Slater orbitals centered on the nucleus of the nitrogen atom. The molecule is predicted to be stable by approximately 0.007 a.u. in both a single- and a five-term treatment, and the ionization potential is found to be 3.94 eV.What is ammonia in aquarium?
Ammonia. Ammonia is formed from the metabolism of protein and is the major waste product of fish. The majority of ammonia from fish is excreted through the gills, with relatively little being lost through urine and feces. Ammonia is also formed as uneaten feed or other organic matter in an aquarium decomposes.How many types of ammonia are there?
three types
Is ammonia a molecule?
The ammonia molecule has a trigonal pyramidal shape, with nitrogen connected to the three hydrogen atoms. The nitrogen atom has a lone electron pair, which makes ammonia a base. NH3 is a polar molecule which readily forms hydrogen bonds, making it highly miscible with water. Chemical properties: Ammonia is a weak base.Can ammonia kill you?
(WOWT) - Anhydrous ammonia is compressed into a clear colorless liquid when used as fertilizer. If you inhale it and it gets in your windpipe and your lungs it will cause burns there that's what will usually kill you - if you inhale concentrated ammonia gas,” said Nebraska Regional Poison Center's Ron Kirschner.Why does ammonia need ice?
Ammonia is stable in plasma for a maximum of 3 hours under these conditions. If sample separation from cells cannot be achieved, the sample should be kept on ice until submission to the laboratory, however ammonia will be less accurate.What is ammonia made of?
Ammonia is made out of one nitrogen and three hydrogen atoms. Its structure is tetrahedral. Ammonia is used in nitric acid production, as a fertilizer, and a cleaning solution. NH3, normally found as a gas, it is caustic and harmful in longterm exposure.What is ammonium chloride in?
Ammonium Chloride. TCC's Ammonium Chloride, NH4Cl, is an inorganic, mildly acidic, white crystalline salt compound that is highly soluble in water. TCC's Ammonium Chloride is mainly used as a nitrogen source in fertilizers, mostly for rice and wheat crops in Asia.What is ammonia found in?
Ammonia is very important to plant, animal, and human life. It is found in water, soil, and air, and is a source of much needed nitrogen for plants and animals. Most of the ammonia in the environment comes from the natural breakdown of manure and dead plants and animals.Is ammonia soluble in water?
Ammonia is NH3 due to the lone pair at the nitrogen the lone pairs in oxygen from the H2O are attracted forming hydrogen bonds consequently being soluble in water . Solubility of a substance in water relies upon on its polar nature. considering water is polar molecule, it dissolves readily polar molecules.Is ammonia a bleach?
Ammonia mainly consists of one nitrogen atom and three hydrogen atoms. Bleach is made from water, caustic soda and chlorine. Ammonia is known as a weak base, while bleach is said to be a strong oxidizing agent. Ammonia consists of one nitrogen atom and three hydrogen atoms.What is the pH of nh4?
about 11
Is ammonium chloride a bleach?
Bleach is a common household name for a solution of sodium hypochlorite and water. Bleach contains NO cleaning agents. Quat is the common name for quaternary ammonium chloride compounds of which there are about 300 varieties all with varying anti-microbial efficacies.How bad is ammonia for you?
Ammonia is corrosive. Exposure to high concentrations of ammonia in air causes immediate burning of the eyes, nose, throat and respiratory tract and can result in blindness, lung damage or death. Inhalation of lower concentrations can cause coughing, and nose and throat irritation.